
Commentary on “Covered TIPS for secondary prophylaxis of variceal bleeding in liver cirrhosis: A systematic review and meta-analysis of randomized controlled trials”
The meta-analysis on Covered TIPS for secondary prophylaxis of variceal bleeding in liver cirrhosis (1) compares covered TIPS with medical treatment consisting of drugs and endoscopic ligation. Previous meta-analyses (2-5) are now obsolete because they included studies which exclusively deployed bare stents and whose medical arms applied treatments that are no more state of the art (e.g., sclerotherapy).
Compared to the bare stents, the use of a covered stent improves TIPS patency and prolongs survival (6-9). Its effect on hepatic encephalopathy (HE) is, however, controversial showing reduced (6) or unchanged (7) rates with covered stents. From a rational basis, better patency of a shunt most likely increases the rate of HE. The findings by Bureau et al. (6) showing lower HE rates with covered stents are irrelevant because their covered stents had smaller diameters than their bare stents (10). With respect to the medical treatment, the use of ligation instead of sclerotherapy improves rebleeding and outcome (11). Previous randomized studies were very heterogeneous providing sclerotherapy alone in 6, ligation alone in 3, sclerotherapy and/or ligation in 1, and endoscopic treatment (sclerotherapy or ligation) plus ß-blocker in three studies (5). Thus, the presently recommended treatment which is ß-blocker plus ligation (12) has not been applied consequently by any of these previous studies.
The present meta-analysis by Qi et al. includes three randomized studies, 2 from Europe (13,14) and 1 from China (15), published in 2015 and 2016 with updated treatment arms using covered stents and drug treatment together with ligation. It shows that “TIPS had a significant benefit of preventing from variceal rebleeding but did not increase the overall survival or risk of hepatic encephalopathy”. The previous (2-5) and the present meta-analyses (1) are similar with respect to rebleeding and survival but differ with respect to HE. In the previous meta-analyses, TIPS significantly increased HE, the reason why TIPS is still recommended as a second line treatment for the prophylaxis of rebleeding (12). In contrast, the present meta-analysis by Qi et al. shows comparable rates of HE assuming “that these findings might influence the future treatment algorithm for the secondary prophylaxis of variceal bleeding in liver cirrhosis”. This assumption is based on the feeling that the balance between rebleeding and HE is now in favour of the TIPS questioning the prime position of the medical treatment.
The European studies (13,14) are very similar with respect to patients and stents (Table 1) with the exception of the nominal stent diameter which was 8 mm in the German (13) and 10 mm in the Netherland study (14). As expected, the smaller stents resulted in a lower reduction of the portosystemic pressure gradient and a lower rate of HE. However, the pressure gradients in the Netherland study before and after TIPS are exceptional low, a fact, which is not explained in the discussion. In contrast to the European studies, the Chinese study (15) implanted 10 mm Fluency stents in patients with advanced cirrhosis, all of them in stage Child-Pugh B and C. In addition, all patients in the Chinese study had some degree of portal vein thrombosis and received post-TIPS anticoagulation with low molecular weight heparin followed by warfarin for 6 months. The different patients and stents may explain the differences in outcome variables of the three studies (Table 2). The decrease in the 2-year survival from Sauerbruch (13) over Holster (14) to Luo (15) may be due to the increasing Child-Pugh score in the latter studies. This may also be a reason for the high rebleeding rates in the Chinese study. However, the presence of portal vein thrombosis, the use of warfarin aiming at an INR between 2 and 3, and the type of stent (Fluency) may also have contributed to the divergent results. The Fluency stent is known for its very high axial recoil which leads to unbending and migration at its ends. All these factors may explain the higher rates of shunt insufficiency (28.7%) and rebleeding (22.2%) in the Chinese TIPS patients. With respect to HE, the Chinese study is unique by showing a higher rate in the medical group. This may in part be due to the high cross over rate of medical patients to TIPS (25%) which is not acknowledged by the “intention-to-treat” (ITT) analysis. This is also true for the Netherland study (14) which reached a significant difference favouring the medical treatment when analysed as “treatment received”. Beneath the many factors mentioned above, the selection of patients with portal vein thrombosis in the Chinese study may be the main factor influencing results. The hemodynamic changes commonly induced by the TIPS (reduction or loss of hepatic portal perfusion) and responsible for TIPS-induced HE, are reduced or abolished in the presence of portal vein thrombosis. Thus, patients with portal vein thrombosis who are free of HE when selected to TIPS, may be candidates with a lower risk of post-TIPS HE. Therefore, from a clinical point of view, the inclusion of the Chinese study may be problematic although heterogeneity was statistically excluded (1). Without this study, the meta-analysis by Qi et al. may approve earlier meta-analyses (2-5) confirming the present recommendation for prophylactic treatment of variceal rebleeding (12). Accordingly, TIPS may remain the second line treatment until additional studies give rise for revision.
Table 1
| Variable | Sauerbruch (13) (n=185) | Holster (14) (n=72) | Luo (15) (n=73) |
|---|---|---|---|
| Child A (%) | 47 | 36 | 0 |
| Child-score | 6.9 | 7.3 | 8.8 |
| Main etiology | Alcohol | Alcohol | Hepatitis B |
| Stent | Viatorr | Viatorr | Fluency |
| Stent nominal diameter (mm) | 8 | 10 | 10 |
| Pressure gradient (mmHg) | |||
| Before TIPS | 22±6 | 13.4±3.3 | 27.5±7.5 |
| After TIPS | 11±5 | 4.4±2.1 | 10.4±3.1 |
| Reduction in pressure gradient (%) | 50 | 67 | 62 |
| Variceal embolization with TIPS | Optional* | 24% | 57% |
*, free decision of interventionalist, exact data not given. TIPS, transjugular intrahepatic portosystemic shunt.
Table 2
| Variable | Sauerbruch (13) (n=185) | Holster (14) (n=72) | Luo (15) (n=73) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| TIPS | Medical | P | TIPS | Medical | P | TIPS | Medical | P | |||
| 2-year survival (%) | 79 | 81 | 0.76 | 72 | 74 | 0.42 | 72.9 | 57.2 | 0.23 | ||
| 2-year rebleeding (%) | 7 | 26 | 0.0018 | 0 | 30 | 0.001 | 22.2 | 57.1 | 0.002 | ||
| 2-year hepatic encephalopathy (%) | 18 | 8 | 0.045 | 38 | 23 | 0.121 | 38.5 | 46.5 | 0.35 | ||
| Cross over rates (%) | 0 | 16 | – | 16 | 17 | – | 5.4 | 25 | – | ||
TIPS, transjugular intrahepatic portosystemic shunt.
Some of the results of the three studies for secondary prophylaxis of rebleeding (13-15) are in contrast with the findings obtained in patients with acute variceal bleeding. A recent multicentre study by García-Pagán (16) compared early placement of a covered TIPS (within 72 hours after admission) with medical treatment in patients at high risk of early rebleeding (Child-Pugh B with active bleeding at endoscopy or Child-Pugh C). Early TIPS had a substantial survival benefit over the medical treatment with a 6-week survival rate of 97% vs. 67%, and a 2-year survival rate of 86% vs. 61% (P<0.001), respectively. The results were confirmed by a post-RCT surveillance study (17) and provided the basis for the Baveno VI consensus which recommends the early placement of a covered TIPS in patients with acute bleeding and a high risk of early rebleeding (12). Moreover, the studies by García-Pagán demonstrated a comparable risk of HE in patients receiving TIPS or medical treatment. This was discussed as being due to the frequent rebleedings in the medical group provoking HE.
The difference in outcomes between early and late (prophylactic) studies may suggest a crucial role of timing of the treatment. Figure 1 depicts the time of randomization and treatment of the acute (16) and prophylactic studies (13,14). The study by Luo (15) is not included because respective data are lacking. If randomization is performed within 24 hours and treatment within 72 hours after bleeding the TIPS had a substantial survival benefit in patients at risk for early rebleeding. If randomization and treatment is performed beyond the acute bleeding phase (4 days) and patients are hemodynamically stabilized, the advantage of the TIPS disappears and survival is comparable between treatment groups. This is true for the early post-acute phase (14) as well as for the following weeks (stratum 1) and months (stratum 2) (13). The reasons for the different survival between groups in early and late (prophylactic) studies may be the different causes of death. Table 3 shows the causes of death of the three studies on secondary prophylaxis (13-15) and the study on acute variceal bleeding (16). Liver failure followed by infection was the most prominent causes of death in the studies for secondary prevention of variceal bleeding. Death due to rebleeding was rare and comparable between groups although rebleeding rates were significantly higher in the medical groups. In contrast, patients with acute bleeding predominantly died from early variceal rebleeding which was successfully prevented by the TIPS (16). Thus, with respect to the causes of death, the early bleeding phase (4 days) clearly differs from the late phase (>4 days) where rebleeding rarely causes death. This is why TIPS does not influence survival in the setting of secondary prophylaxis of rebleeding.
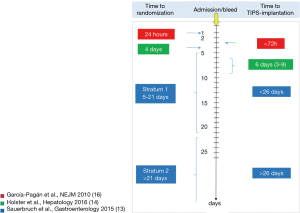
Table 3
| Variable | TIPS | Medical treatment | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Sauerbruch | Holster | Luo | García-Pagán (16) | Sauerbruch | Holster | Luo | García-Pagán (16) | ||
| Overall mortality (%) | 30 | 22 | 32 | 12.5 | 26 | 20 | 47 | 39 | |
| LTX (n) | 5 | 4 | 0 | 2 | 4 | 3 | 0 | 4 | |
| Liver failure (n) | 11 | 3 | 9 | 0 | 9 | 0 | 10 | 2 | |
| Infection (n) | 6 | 2 | 0 | 3 | 8 | 0 | 1 | 4 | |
| Bleeding (n) | 1 | 0 | 1 | 0 | 3 | 3 | 3 | 5 | |
| HCC (n) | 1 | 2 | 1 | 0 | 2 | 2 | 1 | 0 | |
| Technical complications (n) | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Others, multiorgan failure (n) | 7 | 1 | 1 | 1 | 2 | 2 | 2 | 1 | |
TIPS, transjugular intrahepatic portosystemic shunt; LTX, liver transplantation; HCC, hepatocellular carcinoma.
In conclusion, the meta-analysis by Qi et al. (1) may confirm the present algorithm for the secondary prophylaxis of variceal rebleeding with the TIPS being the second line treatment after failure of medical therapy. Patients surviving the acute bleeding tolerate future rebleedings and are more likely to die from other complications of cirrhosis or portal hypertension. In view of similar survival rates, treatment selection (drugs plus ligation or TIPS) may be of secondary importance and should consider individual needs and risks, i.e., severity and frequency of bleedings, risk of HE, additional complications of portal hypertension, rather than a fixed treatment algorithm.
Acknowledgements
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, AME Medical Journal. The article did not undergo external peer review.
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/amj.2017.03.02). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Qi X, Tian Y, Zhang W, et al. Covered TIPS for secondary prophylaxis of variceal bleeding in liver cirrhosis: A systematic review and meta-analysis of randomized controlled trials. Medicine (Baltimore) 2016;95:e5680. [Crossref] [PubMed]
- Luca A, D'Amico G, La Galla R, et al. TIPS for prevention of recurrent bleeding in patients with cirrhosis: meta-analysis of randomized clinical trials. Radiology 1999;212:411-21. [Crossref] [PubMed]
- Papatheodoridis GV, Goulis J, Leandro G, et al. Transjugular intrahepatic portosystemic shunt compared with endoscopic treatment for prevention of variceal rebleeding: A meta-analysis. Hepatology 1999;30:612-22. [Crossref] [PubMed]
- Burroughs AK, Vangeli M. Transjugular intrahepatic portosystemic shunt versus endoscopic therapy: randomized trials for secondary prophylaxis of variceal bleeding: an updated meta-analysis. Scand J Gastroenterol 2002;37:249-52. [Crossref] [PubMed]
- Zheng M, Chen Y, Bai J, et al. Transjugular intrahepatic portosystemic shunt versus endoscopic therapy in the secondary prophylaxis of variceal rebleeding in cirrhotic patients: meta-analysis update. J Clin Gastroenterol 2008;42:507-16. [Crossref] [PubMed]
- Bureau C, Pagan JC, Layrargues GP, et al. Patency of stents covered with polytetrafluoroethylene in patients treated by transjugular intrahepatic portosystemic shunts: long-term results of a randomized multicentre study. Liver Int 2007;27:742-7. [Crossref] [PubMed]
- Perarnau JM, Le Gouge A, Nicolas C, et al. Covered vs. uncovered stents for transjugular intrahepatic portosystemic shunt: A randomized controlled trial. J Hepatol 2014;60:962-8. [Crossref] [PubMed]
- Yang Z, Han G, Wu Q, et al. Patency and clinical outcomes of transjugular intrahepatic portosystemic shunt with polytetrafluoroethylene-covered stents versus bare stents: a meta-analysis. J Gastroenterol Hepatol 2010;25:1718-25. [Crossref] [PubMed]
- Qi X, Guo X, Fan D. A Trend Toward the Improvement of Survival After TIPS by the Use of Covered Stents: A Meta-Analysis of Two Randomized Controlled Trials. Cardiovasc Intervent Radiol 2015;38:1363-4. [Crossref] [PubMed]
- Rössle M, Mullen KD. Long-term patency is expected with covered TIPS stents: this effect may not always be desirable! Hepatology 2004;40:495-7. [Crossref] [PubMed]
- Dai C, Liu WX, Jiang M, et al. Endoscopic variceal ligation compared with endoscopic injection sclerotherapy for treatment of esophageal variceal hemorrhage: a meta-analysis. World J Gastroenterol 2015;21:2534-41. [Crossref] [PubMed]
- de Franchis R, Baveno VI. Faculty. Expanding consensus in portal hypertension: Report of the Baveno VI Consensus Workshop: Stratifying risk and individualizing care for portal hypertension. J Hepatol 2015;63:743-52. [Crossref] [PubMed]
- Sauerbruch T, Mengel M, Dollinger M, et al. Prevention of Rebleeding From Esophageal Varices in Patients With Cirrhosis Receiving Small-Diameter Stents Versus Hemodynamically Controlled Medical Therapy. Gastroenterology 2015;149:660-8.e1. [Crossref] [PubMed]
- Holster IL, Tjwa ET, Moelker A, et al. Covered transjugular intrahepatic portosystemic shunt versus endoscopic therapy + β-blocker for prevention of variceal rebleeding. Hepatology 2016;63:581-9. [Crossref] [PubMed]
- Luo X, Wang Z, Tsauo J, et al. Advanced Cirrhosis Combined with Portal Vein Thrombosis: A Randomized Trial of TIPS versus Endoscopic Band Ligation Plus Propranolol for the Prevention of Recurrent Esophageal Variceal Bleeding. Radiology 2015;276:286-93. [Crossref] [PubMed]
- García-Pagán JC, Caca K, Bureau C, et al. Early use of TIPS in patients with cirrhosis and variceal bleeding. N Engl J Med 2010;362:2370-9. [Crossref] [PubMed]
- Garcia-Pagán JC, Di Pascoli M, Caca K, et al. Use of early-TIPS for high-risk variceal bleeding: results of a post-RCT surveillance study. J Hepatol 2013;58:45-50. [Crossref] [PubMed]
Cite this article as: Rössle M, Schultheiss M. Commentary on “Covered TIPS for secondary prophylaxis of variceal bleeding in liver cirrhosis: A systematic review and meta-analysis of randomized controlled trials”. AME Med J 2017;2:33.

