
Clinical usefulness of automated cellular analysis of synovial fluids: a paradigmatic case report for diagnosing peri-prosthetic infections
Introduction
Bone surgery site infections after osteosynthesis, conventionally known as osteosynthesis-associated infection, is a relative rare but extremely severe complication (1). The cumulative frequency of osteosynthesis-associated infection can be as high as 30% in certain categories of patients, especially those with wound contamination of third-degree open fractures (2). The diagnosis of osteosynthesis-associated infection is frequently challenging, and encompasses an integration of many diagnostic investigations such as laboratory and microbiological testing, diagnostic imaging and histopathology. Notably, recent evidence suggests that the diagnostic performance of periprosthetic tissue cultures is considerably low and may generate a consistent number of false negative test results (i.e., up to one-third) (2). Delayed or inaccurate diagnoses are major determinants of adverse clinical outcomes after osteosynthesis, which may then predispose the patients to severe complications such as retarded bone union and debridement, up to the possibility of amputation (2). In these potential unfavorable scenarios, diagnostic tools enabling rapid rule in or rule out of osteosynthesis-associated infections become pivotal.
The differential diagnosis between infectious and non-infections joint diseases typically entails arthrocentesis with subsequent morphological analysis of synovial fluid, thus including cell count and differential (3). Although this approach is universally validated, there is still uncertainty surrounding the diagnostic cut-offs. A number of leukocytes >2×109/L with as many as 75% polymorphonuclear leukocytes (PMN) is usually diagnostic of an inflammatory condition, whilst a leukocyte count >50×109/L with >85% PMN is suggestive for septic arthritis (4). Unlike these thresholds, the diagnostic cut-offs for diagnosing peri-prosthetic infection remains controversial. The current availability of diagnostic techniques for morphological analysis of synovial fluids is another major drawback. As endorsed by the Clinical and Laboratory Standards Institute (5), optical microscopy remains the gold standard for leukocyte count and differential in synovial fluid. Nevertheless, this technique has many well-known drawbacks, such as poor standardization, high inter-operator variability, long turnaround time, low throughput and need for trained and skilled personnel. Recent evidence has been provided that automated cellular analysis of synovial fluids may represent a valuable perspective for improving both accuracy and timeliness in diagnosing peri-prosthetic infections (6). We describe here a paradigmatic case, documenting the important role of automated morphological analysis of synovial fluids in peri-prosthetic infections diagnostics.
Case presentation
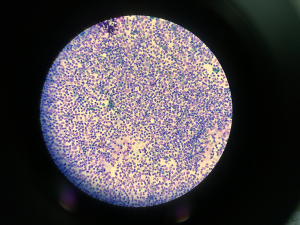
An 18 years old male patient underwent osteosynthesis surgery with a plate and screws for a multi-fragment fracture of his right femur, in December 2000. The patient developed non-union of femoral fracture and serum biomarkers were elevated. Erythrocyte sedimentation rate (ESR) (Roller 20, Alifax, Padova, Italy) was 45 mm/h, whilst C-reactive protein (CRP) (immunoturbidimetric assay on AU480, Beckman Coulter, Brea, CA, USA) was 20 mg/L, respectively. A leukocyte scintigraphy showed an increased uptake at site of pseudarthrosis. The patient underwent additional surgical procedure to remove the metal plate and screws in his right femur. Tissue samples culture for aerobic and anaerobic bacteria were obtained and methicillin-resistant Staphylococcus aureus was isolated from cultured specimens. In February 2003, after multiple antibiotic treatment courses, the patient developed knee joint stiffness and degenerative arthritic changes, and then underwent total knee replacement surgery with a constrained long-stemmed implant due to associated non-union at metaphyseal level. The postoperative period was immediately complicated by appearance of sinus tract in the knee and quadriceps strength deficiency. The patient underwent chronic suppressive antibiotic therapy with minocycline 100 mg bid and trimethoprim/sulfamethoxazole tid. The patient developed extreme knee joint stiffness and ankylosis and intermittent output from the fistula, alternatively suppressed by multiple antibiotic treatment courses. On May 2017, the patient was admitted with severe functional impairment and inability to walk without crutches. Physical examination revealed extensive erythema, swelling at surgical site and pus leakage from a sinus-tract. The patient was afebrile, but laboratory tests revealed an ESR of 32 mm/h and a CRP of 27.8 mg/L. An arthrocentesis was hence performed, followed by white blood cell count (SF-WBC) and differential in synovial fluid. Cellular analysis was carried out using Sysmex XN 2000 body fluid mode (Sysmex Corporation, Kobe, Japan) after addition of hyaluronidase (Sigma Chemical Co., St. Louis, MO, USA) at final concentration of 0.5 mg/mL, as for validated practice (6). The final WBC count was 38×109/L, with 95% PMN, thus compatible with peri-prosthetic infection. The appearance of the synovial fluid on cytospin is shown in Figure 1, confirming the massive amount of PMN already identified by automated cell count and differential in the synovial fluid. The patient underwent additional surgery to remove the knee replacement, accurate debridement and antibiotic impregnated static cement spacer implantation. Three months after the operation, due to infection persistence, the patient underwent another surgery to remove the spacer, replaced by a new static device. In May 2018, considering the severe condition of the surgical area, surgical debridement and removal of the spacer was scheduled with resection arthroplasty. All the tissue samples collected for microbiological testing were positive for Escherichia coli, and Enterococcus faecalis. In June 2018 the patient received another antibiotic impregnated static cement spacer. In November 2018 the patient presented a sinus tract, and underwent another arthrocentesis for physical and microbiological examination of synovial fluid. The leukocyte count and differential in the synovial fluid were again performed with Sysmex XN 2000 body fluid mode (Sysmex Corporation), and revealed the presence of 44.5×109/L cells, with 90% PMN. The synovial fluid was also positive for Van-A Enterococcus faecalis, so that right hip disarticulation was urgently planned. The patient was then discharged and no further complications have been reported so far.
Discussion
Albeit the diagnostic cut-offs remain still to be defined, a recent systematic review and meta-analysis of the literature concluded that an increased leukocyte count in synovial fluid is highly suggestive of peri-prosthetic joint infections, displaying 0.89 sensitivity and 0.86 specificity (7). Notably, the percentage of PMN in synovial fluid had virtually identical diagnostic performance, but a slightly higher area under the curve (i.e., 0.93 versus 0.91). Shortly afterwards, a second independent systematic review and meta-analysis has confirmed that a leukocyte count >3×109/L in synovial fluid is characterized by 0.89 sensitivity and 0.85 specificity for diagnosing peri-prosthetic hip and knee infections (8). Even in such case the diagnostic performance of percentage PMN in synovial fluid was comparable to that of the leukocyte count, exhibiting 0.88 sensitivity and 0.82 specificity. Irrespective of the final cut-offs, which are usually comprised between >1.7×109/L and >4.5×109/L, the leukocyte count in synovial fluid shall hence be regarded now as a cornerstone in peri-prosthetic infections diagnostics, thus reinforcing the crucial role played by laboratory diagnostics in modern medicine, always at the center of both the diagnostic reasoning and clinical decision making (9). Despite optical microscopic analysis is still considered the reference method for cell analysis in synovial fluid, the new generation of hematological analyzers is characterized by specific features enabling rapid, accurate and reliable measures. Therefore, their use in routine laboratory practice is increasingly described as a valid alternative to the manual technique, as also shown in this paradigmatic case report of peri-prosthetic infection.
Conclusions
In conclusions, this case provides clear evidence that automated leukocyte count and differential in synovial fluid shall now be considered an unavoidable part of the diagnostic approach to patients with suspected peri-prosthetic infections.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/amj.2019.05.03). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained for publication of this Case Report and any accompanying.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Fang C, Wong TM, Lau TW, et al. Infection after fracture osteosynthesis - Part I. J Orthop Surg (Hong Kong) 2017;25:2309499017692712 [PubMed]
- Yano MH, Klautau GB, da Silva CB, et al. Improved diagnosis of infection associated with osteosynthesis by use of sonication of fracture fixation implants. J Clin Microbiol 2014;52:4176-82. [Crossref] [PubMed]
- Margaretten ME, Kohlwes J, Moore D, et al. Does this adult patient have septic arthritis? JAMA 2007;297:1478-88. [Crossref] [PubMed]
- Shah RP, Plummer DR, Moric M, et al. Diagnosing Infection in the Setting of Periprosthetic Fractures. J Arthroplasty 2016;31:140-3. [Crossref] [PubMed]
- Clinical and Laboratory Standard Institute. Body Fluid Analysis for Cellular Composition; Approved Guidelines (CLSI document H56-A). Wayne, PA, 2006.
- Seghezzi M, Buoro S, Manenti B, et al. Optimization of Cellular analysis of Synovial Fluids by optical microscopy and automated count using the Sysmex XN Body Fluid Mode. Clin Chim Acta 2016;462:41-8. [Crossref] [PubMed]
- Lee YS, Koo KH, Kim HJ, et al. Synovial Fluid Biomarkers for the Diagnosis of Periprosthetic Joint Infection: A Systematic Review and Meta-Analysis. J Bone Joint Surg Am 2017;99:2077-84. [Crossref] [PubMed]
- De Fine M, Giavaresi G, Fini M, et al. The role of synovial fluid analysis in the detection of periprosthetic hip and knee infections: a systematic review and meta-analysis. Int Orthop 2018;42:983-94. [Crossref] [PubMed]
- Plebani M, Laposata M, Lippi G. A manifesto for the future of laboratory medicine professionals. Clin Chim Acta 2019;489:49-52. [Crossref] [PubMed]
Cite this article as: Baldini A, Balboni F, Balato G, Buoro S, Pezzati P, Quercioli M, Strigelli V, De Franco C, De Matteo V, Lippi G. Clinical usefulness of automated cellular analysis of synovial fluids: a paradigmatic case report for diagnosing peri-prosthetic infections. AME Med J 2019;4:29.

