
Management of malignant airway obstruction
Introduction
The term “Malignant Central Airway Obstruction” (MCAO) refers to any malignant, mechanical, obstructive process that impedes the airflow within the central airways (trachea, main-stem bronchi, and right bronchus intermedius). MCAO represents a substantial source of morbidity and mortality with a significant impact on quality of life (1,2).
Malignant obstruction of the central airways usually presents late in the course of the disease, and most individuals have a limited life expectancy. It creates significant physiological and psychological distress in patients and occasionally, may present as imminent arrest from suffocation. Traditional modalities of treatment, such as chemotherapy and external beam radiation are ineffective for the acute management of severe MCAO, not only in restoring oxygenation and ventilation, but also in palliating symptoms. Multiple highly effective interventional pulmonology techniques are now available for the management of malignant obstruction of the central airways.
Prevalence
The actual incidence and prevalence of MCAO are unknown, though an estimated 80,000 cases of malignant airway obstruction are treated annually in the US (3). Unfortunately, it is not infrequently seen in clinical practice, especially in advanced malignancies. Approximately 20% to 30% of patients with lung cancer may develop a complication related to central airway obstruction, such as dyspnea, atelectasis, hypoxemia, hemoptysis, post-obstructive pneumonia or respiratory distress. About 40% of lung cancer-related deaths result from complications due to advanced loco-regional disease (2,4-7).
Lung cancer remains the leading cause of cancer death in both men and women in the US. Given the contemporary epidemiology of lung cancer, the incidence and prevalence of malignant CAO may be increasing due to the rising number of lung cancer patients developing complications related to endobronchial disease (2).
Etiology
Malignant obstruction of the central airways may develop due to a primary intraluminal malignancy, extension of an adjacent tumor with airway invasion, metastatic endo-luminal disease, or extrinsic compression from a contiguous malignant process (8). The most common cause of MCAO is direct extension and invasion from an adjacent tumor, often bronchogenic carcinoma (2,9). Squamous cell carcinoma accounts for more than half of the non-small-cell lung cancer (NSCLC)-related MCAO (10,11). Other tumors commonly associated with adjacent endobronchial invasion include esophageal, laryngeal, and thyroid malignancies.
Primary malignancies of the airways are rare. Squamous cell carcinoma and adenoid-cystic carcinoma account for two-thirds of all primary tracheal malignant tumors (12-16). Distal to the main carina, carcinoid tumors account for many primary airway malignancies, typically presenting as discrete central endobronchial lesions (16,17).
Endoluminal metastases from distant malignancies are relatively uncommon, with the reported incidence ranging widely from 2% to 50%. The considerable variation is likely related to the variable definitions used; the incidence is much lower when only distant tumors that directly metastasize to the airways are considered (18,19). In one autopsy series of patients with solid tumors, metastatic disease to the central airways occurred in only 2% of the cases (20). However, the incidence of extra-thoracic endoluminal metastases presenting as symptomatic MCAO is not known. A wide variety of tumors that metastasize to the airways have been described, including breast, colorectal, renal cell, and thyroid carcinomas (2,18,19).
Types/classification (Figure 1)
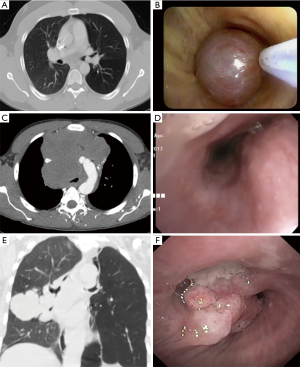
Three basic types of MCAO have been widely described:
- Intrinsic or endoluminal obstruction: Airway lumen compromised purely by an endo-bronchial/tracheal obstructive tumor.
- Extrinsic or extraluminal obstruction: Airway compressed by an extra-bronchial/tracheal malignant process.
- Mixed obstruction: A combination of intraluminal and extraluminal airway obstruction.
Clinical presentation
MCAO not only carries a very poor prognosis if left untreated, but also results in significant daily life disturbances. Patients are often quite symptomatic with severely impaired quality of life. The symptomatology of MCAO is diverse and nonspecific, with shortness of breath and cough being the most commonly reported symptoms. Other frequently reported symptoms include hemoptysis, hoarseness, chest discomfort, orthopnea, and dysphagia (21). Patients may be asymptomatic if the obstruction is mild. Conversely, even in mild obstructions during an acute respiratory infection, the concomitant edema and buildup of secretions may decrease the airway lumen at the obstructive site, leading to significant symptoms. Shortness of breath is usually a late sign that may develop at rest or on exertion. It is occasionally positional with dyspnea occurring in the supine position due to airway compression by large intrathoracic tumors. Dyspnea characteristically is persistent and not responsive to bronchodilators. The level of shortness of breath does not always correlate with the amount of airway obstruction. Cough is usually chronic, persistent, and may be dry or productive of purulent sputum. Hemoptysis is common. Though most studies report a mild to moderate degree, massive hemoptysis may occur. Dysphagia may be present in patients with large airway malignancies causing esophageal compression, or with esophageal malignancies with endobronchial invasion (2,22,23).
On chest auscultation, stridor, wheezing, localized crackles, or frank consolidation may be encountered, depending on the location and size of the obstruction-related atelectasis. The location of wheezing does not always follow the site of the airflow obstruction and may be heard over the trachea or lung fields (24). Unilateral wheezing suggests obstruction distal to the carina. Wheezing related to MCAO is generally not responsive to bronchodilators (23,25).
Work-up/imaging/diagnostic bronchoscopy
The diagnosis of MCAO can be challenging. It requires a high clinical suspicion and is usually based on a combination of past medical historic factors and characteristic findings on physical exam, as well as physiologic, imaging, and endoscopic studies. A detailed clinical history is crucial for diagnosis. The past medical history must be carefully reviewed, and previous medical records obtained (26). Any active or previous malignant diseases should be thoroughly researched, as most patients with MCAO have an end-stage primary tumor or a recurrence following prior surgical or chemo-radiation regimens (27). A family history of malignancy should also be also noted.
A plain chest X-ray should be obtained in every patient suspected to have MCAO. Although it is infrequently diagnostic and has a low sensitivity detecting abnormalities of the trachea and main bronchi, it may reveal obvious pathologies such as tracheal deviation from an adjacent lesion, lobar atelectasis, or complete lung collapse (28,29).
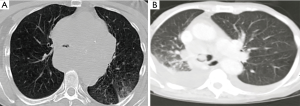
Computed tomography (CT) of the thorax is the most accurate noninvasive method of airway evaluation, allowing diagnosis and treatment planning in MCAO. CT as an imaging modality is superior to chest X-ray in detecting tracheal and main bronchi abnormalities, with a reported sensitivity of 97% (28). A CT of the chest helps determine the type of obstructive lesion (intraluminal, extrinsic, or mixed), the patency of the airway distal to the obstruction, and important lesion characteristics, including length, diameter, and relationship to nearby structures such as vessels. Intratracheal or intrabronchial masses and signs of extrinsic tracheal or bronchi compression may be evident (Figure 2) (30,31). Post-obstructive pneumonia, atelectasis, and lobar collapses may also be visible. CT of the chest with multi-planar reconstructions has a reported 93% sensitivity, 100% specificity, and 94% accuracy in demonstrating the site and degree of tracheal and main bronchi stenosis (32).
Bronchoscopy, either flexible or rigid, is the most specific and sensitive test for MCAO and is used for the assessment, diagnosis and treatment, often simultaneously during the initial procedure (33,34). Direct visualization via bronchoscopy can provide information on the location and morphology of the lesion, the amount of intraluminal disease, the presence of extraluminal compression, and the diameter and length of the lesion. It also allows evaluation of the surrounding tissue, particularly the airway distal to the obstruction. Additionally, tissue can be obtained for pathologic diagnosis and staging if necessary. Diagnostic flexible bronchoscopy has several detriments and should be performed with caution. It does not provide a complete evaluation of extraluminal disease or the viability of the airways distal to the obstruction. It may also be difficult and potentially dangerous in severe obstruction, as the bronchoscope itself will further obstruct the already narrow lumen, particularly if secretions, edema, or bleeding occur (27). The moderate sedation required during the procedure may decrease ventilation and relax the respiratory muscles, creating a potentially unstable airway, especially with anterior mediastinal tumors. For these reasons, an interventional pulmonology team is crucial when diagnostic flexible bronchoscopy is performed in MCAO. In cases of severe obstruction presenting with impending respiratory failure, diagnostic studies including preliminary flexible bronchoscopy may not be possible, and a rigid bronchoscopy should be performed immediately.
Management
The management of MCAO is challenging and requires a multidisciplinary team approach with the involvement of a pulmonologist, medical and radiation oncologist, anesthesiologist, ENT specialist, thoracic surgeon, and interventional pulmonologist. Surgery is often not indicated due to the advanced disease state or the patient’s comorbidities. Chemotherapy has inconsistent and delayed beneficial effects and radiotherapy often yields suboptimal results, with delayed atelectasis resolution obtained in only half of the cases (35,36).
Management is mostly dependent on the initial presentation. Approximately 40% of the interventions performed for airway obstruction are done either on an urgent or emergent basis (37). Significant malignant airway obstruction presenting with severe respiratory distress requires immediate action to promptly and effectively re-establish and secure the airway as well as to relieve the obstruction (27). Thus, in unstable patients presenting with severe tracheal or bronchial obstruction and impending respiratory failure, initial stabilization should be focused on establishing a secure airway. This may require endotracheal intubation or urgent rigid bronchoscopy. If an interventional pulmonology team is not available, patient transfer to a specialized center should be considered after initial stabilization.
Malignant airway obstruction often presents in a late stage where curative surgical resection is not an option due to the extent of malignant disease at presentation and the existence of comorbid medical conditions that render many patients unsuitable for surgery (34,38). However, when feasible, resection of the malignant process is the treatment of choice. Retrospective studies suggest that therapeutic bronchoscopy can be used as a complementary tool in the management of MCAO prior to curative surgery (11).
Additionally, few cases have described non-resectable lung cancers that have become operable following interventional bronchoscopic therapies (10,39). Unfortunately, the majority of lung cancer patients present in advanced stages, either stage III or stage IV (40), therefore management is focused primarily on symptom palliation and quality of life improvement.
In patients with inoperable tumors of the central airway, restoration of airway patency provides palliation and may prolong life, especially in cases presenting with impending respiratory failure (33). No established guideline exists for the management of MCAO. Several techniques are available for relieving the airway obstruction; the choice of which to use depends on the obstruction type, the patient’s clinical condition, equipment availability and treating physician’s expertise. Interventional bronchoscopy procedures may be indicated prior to chemotherapy or radiation therapy (or when such treatment fails) and have been shown to improve dyspnea as well as mechanical ventilation liberation rates, thus increasing quality of life (10). Consequently, the 2013 American College of Chest Physicians (ACCP) evidence-based clinical practice guidelines recommend that in patients with inoperable lung cancer and symptomatic airway obstruction, therapeutic bronchoscopy with mechanical or thermal ablation, brachytherapy, or stent placement, should be offered with the aim of improving dyspnea, cough, hemoptysis, and quality of life (41).
Endoscopic management
Multiple and often complementary techniques exist in the armamentarium of interventional pulmonology for the management of MCAO. Selection of the appropriate approach depends on several factors, including the acuity of the presentation, the underlying cause, the type of lesion, the stability of the patient, the patient’s general, cardiac, and pulmonary status, the quality of life, the overall prognosis, the physician expertise, and the technology available (42-44). Multimodal management utilizing various endoscopic techniques is generally used (Figure 3) (45).
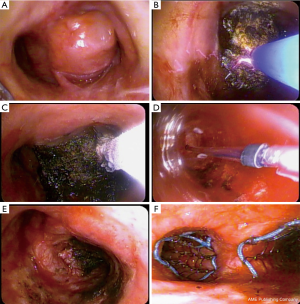
Subsequently, we will describe the more frequently used procedures.
Rigid and flexible bronchoscopy
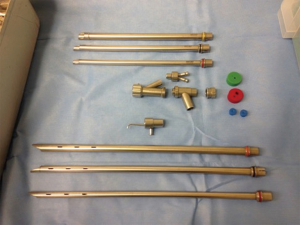
Bronchoscopic therapy, performed via flexible or rigid bronchoscopy, results in an improvement in symptoms, quality of life, and survival. Both techniques may be complementary. Rigid bronchoscopy offers excellent airway control. It is a safe and highly effective way of securing the airway while providing the capability to ventilate and oxygenate during diagnostic and therapeutic airway procedures (34,45-47). Rigid bronchoscopy is considered by experts the modality of choice in patients with impending respiratory failure. It requires general anesthesia and an operating theater. Contraindications include those related to anesthesia as well as complex anatomy of the neck and mandible (i.e., unstable cervical spine, oral or maxillofacial trauma) (46). Complications are infrequent with the most common being a sore throat after the procedure. Other reported complications include injury to the teeth, gums or lips, tracheal or bronchial tears, severe bleeding, and hypoxemia-induced cardiac ischemia and arrhythmia. The overall mortality related to rigid bronchoscopy is as low as 0.4% (45-48). Over the past several years, various improvements have occurred to the design of the rigid bronchoscope, including the ability to measure inspiratory and expiratory pressures as well as oxygen and carbon dioxide concentrations, creating a more versatile tool (Figure 4) (49,50). Rigid bronchoscopy requires specialized training and as such, is underutilized in the United States due to the scarcity of training environments. Rigid bronchoscopy training is offered in 4.4% of all pulmonary medicine programs and only 31.3% of pulmonary programs with an interventional pulmonology service (48).
Thermal techniques
All thermal endoscopic airway interventions can be used with either a rigid or a flexible bronchoscope.
Light amplification of stimulated emission of radiation (LASER)
Laser photoresection refers to the application of LASER energy to produce thermal, photodynamic, and electromagnetic changes in tissues. Numerous types of LASER are available, including neodymium: yttrium-aluminum-garnet (Nd:YAG), carbon dioxide (CO2), and neodymium: yttrium-aluminum-perovskite (Nd:YAP), among others. Nd:YAG LASER is probably the most widely used for endobronchial disease. Nd:YAG is a noncontact or contact technique in which we apply thermal energy to the airway tissue for the relief of malignant obstruction. The LASER thermal technique can be used in emergent situations and is an excellent instrument for rapid endoluminal debulking, with a reported rate of 83% to 93% of lumen re-establishment and a 63% to 94% rate of symptom palliation. Excellent knowledge of anatomy is essential to avoid complications, such as major vessel perforation. As the depth of tissue destruction cannot be precisely assessed just by the appearance of the tissue surface, extreme caution should be taken to direct the laser beam parallel to the bronchial wall to avoid damage. To minimize movement, general anesthesia with neuromuscular relaxant is recommended (e.g., coughing). LASER, as with other thermal techniques, is not indicated in isolated extrinsic MCAO. Complications are rare in experienced hands and are estimated to be <3%. Such complications include perforation (of airways, esophagus, or vessels), cardiac arrhythmias, pneumothorax, hemorrhage, hypoxemia, myocardial infarction, air embolism (secondary to gas leaving the probe tip under pressure and crossing the mucosal membranes into the blood vessels), and endobronchial ignition. The fraction of inspired oxygen (FiO2) should remain less than 40% during the procedure to avoid endobronchial fire. Currently, no randomized trials exist to compare Nd:YAG LASER with other forms of MCAO management (i.e., electrocautery or cryotherapy), but several retrospective studies have shown successful outcomes using Nd:YAG LASER by itself or in combined modality therapy (2,9,26,27,43,45,47,51-56).
Electrocautery
Electrocautery or electrosurgery refers to the use of an electric current to heat and destroy tissue. It can be a contact or noncontact technique in which a high-frequency alternating electric current is delivered to the obstructive tissue for the relief of a malignant obstruction. The effect on the tissue depends on the power used, the application time, the contact surface area, and the tissue type. The heat generated by the electric current is proportionally related to tissue resistance, and inversely related to tissue vascularity and moisture content. Depending on the type of accessory use, the Power (W) settings range from 10–40 W. Electrocautery may be used in emergent situations and is an excellent tool for rapid endoluminal debulking, with a reported rate of lumen re-establishment of approximately >90%, and symptomatic improvement in more than 90% of patients. A variety of tools exist to apply electrocautery, via either rigid or flexible bronchoscopy. For the flexible bronchoscope, electrocautery snares, knives, blunt probes, and hot forceps are available. Electrocautery should be avoided in pure extrinsic airway compression. In patients with pacemakers or automatic implantable cardioverter/defibrillators (AICDs), due to the potential for dysrhythmias or device malfunction, caution is recommended, and the device should be turned off whenever possible and clinically indicated. Complications are rare, estimated between 2–5%, and include hemorrhage, endobronchial ignition, electric shock to the operator, and airway perforation. Loss of efficacy can occur with bleeding due to the diffusion of the current across a larger surface area. The FiO2 must be below 40% to avoid an airway fire (2,42,43,47,54,56-59).
Argon plasma coagulation (APC)
APC is a non-contact mode of tissue electrocoagulation in which ionized argon gas is used to conduct electric current to the obstructive tissue for the management of MCAO. APC is used more frequently as an alternative to LASER therapy and electrosurgery, as it is an excellent tool for photocoagulation (hemostasis) with a rate of luminal restoration of approximately 91% and a remarkable safety profile. Three types of flexible APC probes are available for different indications: 0-degree or head-on, radial and lateral. APC can target lateral lesions or those at sharp angles to the probe. APC can be used in a forced or pulse mode, and the power setting may be set between 10–30 W. However, APC does not cause tumor vaporization, thus requiring other modalities for the debulking of large tumors. After applying APC to an endoluminal tumor, the resultant eschar and debris must be removed with suction, forceps, or cryoadhesion. As with LASER and electrocautery, APC is contraindicated in extrinsic airway compression, and the same principle of caution applies for patients with pacemakers or AICDs. A complication rate of <1% has been reported. Complications include hemorrhage, airway perforation, airway stenosis, endobronchial ignition, and air embolism from the argon gas. The recommended flow of gas is from 0.3 to 0.8 LPM to avoid an embolism event. Also, as with the previously described thermal modalities, the FiO2 must be below 40% to avoid an airway fire (2,43,47,54-56,59,60).
Cryotherapy
Cryotherapy utilizes extreme cold to destroy tissue by rapid freezing and slow thawing cycles. Cryotherapy for endobronchial tumor management was first described in 1968 (61). It is a contact technique with cryogen (i.e., nitric oxide) application to the obstructive tissue. Most of the effects of cryotherapy start several hours after treatment, therefore historically the use of cryotherapy in malignant airway obstructions was limited to non-acute obstruction.
It is also important to remember that tissue necrosis and sloughing at 1–2 weeks, requires removal of debris to achieve the desired effect (43,45,56,62,63). However, with the use of cryoadhesion, debulking, and recanalization, the role of the cryoprobe in malignant endoluminal airway obstruction has expanded. Case reports (64) and retrospective studies have shown that the use of cryotherapy for cryorecanalization can achieve an immediate treatment effect with an adequate safety and efficacy profile (61). A large retrospective study of 225 cases found a 91% success rate using the flexible cryoprobe for cryo-recanalization of malignant stenosis (65). Cryotherapy is a safe procedure with few and relatively minor complications. In the published literature, the most important complication of cryorecanalization is bleeding. Experienced users of cryo-debridement recommend the use of other techniques for hemostasis (i.e., APC or electrocautery) in as many as 8% to 10% of cases (61,65-67). One of the main advantages of using cryotherapy up front in MCAO debulking is the ability to diagnose (cryobiopsies) and debulk with recanalization as the initial diagnostic and therapeutic procedure.
Non-thermal techniques
Mechanical debulking by rigid bronchoscopy
The use of isolated mechanical debulking for treatment of MCAO has been previously described.
A retrospective study has shown that rigid bronchoscopy and mechanical debulking as a sole therapy is safe and successful in up to 83% of cases of central airway tumors (68). In rigid debulking, the distal end of the rigid tracheoscope or bronchoscope acts as a corkscrew for dilating the stenotic segment, or as an apple corer penetrating through large obstructive tumors. The barrel of the bronchoscope can be used simultaneously to tamponade bleeding lesions while debulking the tumor. Large forceps may be introduced through the bronchoscope to assist in the mechanical debridement of large tumors. A flexible bronchoscope may be used during rigid bronchoscopy to facilitate tissue debridement in angulated or distal airways. These techniques, although still commonly used, should be reserved for the most severe cases. Complications with mechanical debulking range from 1–20%, and include pneumothorax, hemoptysis, and pneumonia (69).
Airway dilation
Dilation of airway obstructions due to intrinsic, extrinsic or mixed MCAO may be achieved with insertion of the barrel of a rigid bronchoscope or with balloon dilation. Rigid bronchoscopic airway dilation can be used in emergent situations, as rapid recanalization can be achieved as described above (33,46). Balloon dilatation or bronchoplasty (BBP) can be performed during rigid or flexible bronchoscopy with or without fluoroscopy and involves the use of increasingly larger diameter balloons filled with saline maintained in position for 15–60 s to dilate the airway. This induces less mucosal trauma and subsequent granulation tissue formation than rigid dilation. BBP results in an immediate improvement in extrinsic and intrinsic MCAO in up to 79% of patients and often used for airway dilation before stenting. As its effects are not long-lasting, BBP dilation is frequently combined with other therapies such as laser resection, radiation therapy, or stenting. Complications include stenosis recurrence, pain, mediastinitis, and bleeding, as well as airway tearing or rupture with subsequent pneumothorax or pneumomediastinum (70,71).
Microdebrider
The microdebrider is a powered rotating blade use to resect endoluminal obstructions. Simultaneous suction facilitates rapid removal of blood and debris with minimal trauma to the airway. The microdebrider blade may be smooth or serrated and comes in 2 lengths: 37 cm, which is used to access lesions of the trachea and the most proximal main bronchi, and 45 cm, used for more distal lesions. The usual speed of the blade is between 1,000 and 2,000 rpm. The microdebrider represents an alternative therapeutic option in patients with poor pulmonary reserve as there is no need to decrease the FiO2 during tumor debulking. Additional techniques such as electrocautery may be necessary to achieve hemostasis in up to 35% of patients. Retrospective studies have found that the microdebrider is safe and effective in the management of MCAO (72-74).
Airway stents
Airway stents are prostheses of various materials used to support and maintain patency of the airway. Two types of stents (silicone and metallic/hybrid) are frequently used in the management of MCAO. Airway stents are best suited for extrinsic malignant compression. They are sometimes used to maintain airway patency after intrinsic or mixed endobronchial tumor ablation, or in cases of persistent airway narrowing. Stents deliver immediate and durable palliation, with symptomatic relief achieved in up to 84% of patients. Tracheobronchial stents improve quality of life and survival in patients with advanced malignant obstruction. When used for prolonged periods of time, stents may develop significant complications. Reported complications include migration, infection, granulation tissue formation, halitosis, stent fracture, metal fatigue, perforation of vessels and airway wall, mucosal tears, and obstruction of lobar orifices. Experts recommend a “stent alert” card to be given to every patient. It should specify the type and size of the stent, the location, and the appropriate size of the endotracheal tube to be used if emergency intubation became necessary.
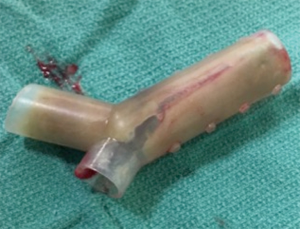
Silicone stents (i.e., Dumon bronchial stent, Dumon-Y stent, Polyflex stent) are placed under rigid bronchoscopy and have several benefits including varying degree of firmness and flexibility to simultaneously mold to the airway while resisting external compression. They are relatively inexpensive and are reasonably well-tolerated (75). Worldwide silicone stents are the most commonly used stents (40). The Dumon silicone stent has studs on the surface to decrease the migration rate. They can be customized to the patient needs prior to insertion (i.e., cut to the desired length, create an orifice for a lobar bronchus) One significant advantage of the silicone stents is the fact that they can be removed with ease. The Dumon-Y silicone stent is particularly useful in malignant diseases involving the carina and the mainstem bronchi. They do, however, have high rates of migration and obstruction as granulation tissue forms at the stent-ends and mucous secretion clearance is impaired (Figure 5).
Self-expanding metallic stents are relatively easy to deploy without the need of rigid bronchoscopy and conform better to disrupted airways. They have better internal-to external diameter ratios and therefore result in larger airway lumens overall. Unfortunately, tumor and granulation tissue often grow through the mesh, making purely metal stents challenging to remove. Complications such as airway and vessel perforation have been reported (Figure 6) (2,6,43,54,75-81).

Hybrid stents (i.e., Ultraflex stent, Aero stent), in theory, combine the qualities of silicone and metallic stents. The covered hybrid stents have the advantage of creating a mechanical barrier, preventing tumor ingrowth and are in general easier to manipulate. Some of these stents have a small proximal or distal loop allowing for partial collapse and subsequent removal or reposition. Others have anti-migration struts which sink into the mucosa to prevent migration, causing significant mucosal injury, particularly at the time of removal (75).
Photodynamic therapy (PDT)
In PDT, a light of specific wavelength [from a potassium titanyl phosphate (KTP) laser] is applied to the obstructive lesion via a flexible bronchoscope 48 to 72 hours after the systemic injection of a photosensitizing drug such as dihematoporphyrin ester (DHE). This leads to a phototoxic reaction and tumor destruction as the photosensitizing drug is preferentially taken up by malignant cells. Immediately and up to 48 hours after the procedure, bronchoscopic toilet (cleaning and debulking of the area to remove tumor debris, retained secretions, and sloughed mucosa) is performed to establish airway patency and assess the necessity of further treatment. PDT is indicated in the palliative treatment of CAO without acute dyspnea and is particularly useful in distal obstructions due to malignant endobronchial masses with minimal extrinsic airway compression. Due to the delayed response of treatment, PDT should not be used in the emergent management of acute, severe MCAO. The most common complication associated with PDT is skin photosensitivity. Patients receiving PDT should be advised to avoid sun exposure for 4–6 weeks after the procedure. Other complications include local airway edema, strictures, hemorrhage, and fistulae formation, although PDT has a low risk of airway perforation. The effects of PDT are relatively long-lasting and have been shown to palliate airway obstruction in 80% of patients (2,42,43,47,54).
Radiation (brachytherapy) endoscopic airway interventions
Endobronchial delivery of radiation is achieved via the placement of a radioactive substance (i.e., iridium-192) directly into or near the airway tumor using a flexible bronchoscope. This results in tissue destruction through DNA mutations leading to cell apoptosis. Brachytherapy is indicated in the palliation of symptoms (particularly dyspnea, cough, and hemoptysis) related to airway obstruction. High-dose endobronchial brachytherapy is also successful in the treatment of excessive granulation tissue formation as a complication of airway stenting. As brachytherapy takes up to 3 weeks to be effective, it should not be used in the emergent management of acute, severe MCAO. The effects of brachytherapy are long-lasting, with a reported rate of lumen restoration of 78% to 85% and symptom relief of 69% to 93%. An advantage of brachytherapy is that it can be used for tumors in areas not accessible to other treatment modalities (e.g., the upper lobe bronchi and segmental bronchi). Brachytherapy is delivered via low-dose-rate (LDR) or high-dose-rate (HDR) endobronchial methods. High-dose endobronchial brachytherapy delivers higher radiation doses with less time per fraction, thus permitting outpatient therapy. Brachytherapy can be used in combination with other techniques such as thermal therapy or external beam radiation, with which it has synergistic effects. Complications of brachytherapy include hemorrhage, mediastinal fistula formation, arrhythmias, hypotension, bronchospasm, bronchial stenosis or necrosis, and radiation bronchitis (45,47,54,63).
Outcome/prognosis
In the vast majority of cases, malignant airway obstruction is not curable, and the approach is aimed at the palliation of symptoms (e.g., dyspnea, cough, hemoptysis). Patients and family members should be well aware of the palliative nature of these efforts. The survival of patients with untreated malignant CAO is generally poor and ranges from 1 to 2 months (82). Furthermore, their quality of life is extremely poor, and they may die with asphyxia or on mechanical ventilation. Several risk factors for decreased survival in cases of MCAO have been described, including a high American Society of Anesthesiologists (ASA) score, non-squamous cell histology, and previously untreated metastatic tumors (36). In patients with malignant airway obstruction, studies have shown that multimodal therapy and stent insertion improves quality of life when compared with other approaches.
A prospective study on patients with MCAO due to advanced or recurrent lung cancer who underwent LASER ablation demonstrated significant improvement in both objective and subjective measures of quality of life (83). Two retrospective studies showed improved palliation and improved survival after airway stenting in advanced lung cancer (79,82). Additionally, a recent prospective study showed that therapeutic bronchoscopy for MCAO significantly improved spirometry values (i.e., FVC, FEV1), quality of life scores and overall survival (84). Chhajed et al. (6) demonstrated that patients presenting with MCAO who had received interventional bronchoscopic therapy prior to oncological treatment, had the same expected survival as those patients presenting in the similar stage without MCAO.
A recent report from the ACCP multicenter registry study of therapeutic bronchoscopy for MCAO showed a very high technical success rate of more than 90%. The highest success rates were associated with stent placement and endobronchial obstruction. A 48% clinical improvement in dyspnea was reported, with 42% improvement in health-related quality of life scores. A more meaningful improvement was seen in those patients who had greater dyspnea at baseline (85).
An overall complication rate of 3.9% (range, 0.9–11.7%) was seen after therapeutic bronchoscopy for MCAO. The risk factors identified for complications included emergent/urgent procedures, an ASA score >3, redo-therapeutic bronchoscopy and moderate sedation. A 14.8% 30-day mortality was described (86).
Conclusions
In summary, MCAO is an important disease entity which significantly impacts a patient’s quality of life and can determine candidacy for systemic or surgical therapies. There are many minimally invasive bronchoscopic interventions which can be used to relieve MCAO, resulting in rapid relief of symptoms, even in acutely ill patients. Current modalities include a variety of thermal techniques, cryotherapy, mechanical debulking, airway dilation, and airway stent placement. Delayed therapies such as brachytherapy and photodynamic therapy are very useful in select cases. Thorough working knowledge of the risks and benefits of each modality is critical when individualizing a patient’s treatment plan. A team of experts including interventional pulmonologists and thoracic surgeons should be involved in these cases.
Acknowledgements
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, AME Medical Journal for the series “Management of Complex Airway and Pleural Diseases”. The article has undergone external peer review.
Conflicts of Interest: The authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/amj.2018.11.06). The series “Management of Complex Airway and Pleural Diseases” was commissioned by the editorial office without any funding or sponsorship. Dr. Folch served as the unpaid Guest Editor of the series. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Rafanan AL, Mehta AC. Role of bronchoscopy in lung cancer. Semin Respir Crit Care Med 2000;21:405-20. [Crossref] [PubMed]
- Ernst A, Feller-Kopman D, Becker HD, et al. Central airway obstruction. Am J Respir Crit Care Med 2004;169:1278-97. [Crossref] [PubMed]
- Ernst A, Herth FJ. Principles and Practice of Interventional Pulmonology. S.l.: Springer-Verlag New York; 2013.
- Dutau H, Toutblanc B, Lamb C, et al. Use of the Dumon Y-stent in the management of malignant disease involving the carina: a retrospective review of 86 patients. Chest 2004;126:951-8. [Crossref] [PubMed]
- Miyazawa T, Miyazu Y, Iwamoto Y, et al. Stenting at the flow-limiting segment in tracheobronchial stenosis due to lung cancer. Am J Respir Crit Care Med 2004;169:1096-102. [Crossref] [PubMed]
- Chhajed PN, Baty F, Pless M, et al. Outcome of treated advanced non-small cell lung cancer with and without central airway obstruction. Chest 2006;130:1803-7. [Crossref] [PubMed]
- Guibert N, Mazieres J, Marquette CH, et al. Integration of interventional bronchoscopy in the management of lung cancer. Eur Respir Rev 2015;24:378-91. [Crossref] [PubMed]
- Davis MP. Supportive oncology. Philadelphia, PA: Elsevier/Saunders, 2011.
- Beamis JF Jr. Interventional pulmonology techniques for treating malignant large airway obstruction: an update. Curr Opin Pulm Med 2005;11:292-5. [Crossref] [PubMed]
- Cosano Povedano A, Muñoz Cabrera L, Cosano Povedano FJ, et al. Endoscopic treatment of central airway stenosis: five years' experience. Arch Bronconeumol 2005;41:322-7. [PubMed]
- Chhajed PN, Eberhardt R, Dienemann H, et al. Therapeutic bronchoscopy interventions before surgical resection of lung cancer. Ann Thorac Surg 2006;81:1839-43. [Crossref] [PubMed]
- Maziak DE, Todd TR, Keshavjee SH, et al. Adenoid cystic carcinoma of the airway: thirty-two-year experience. J Thorac Cardiovasc Surg 1996;112:1522-31. [Crossref] [PubMed]
- Mathisen DJ. Primary tracheal tumor management. Surg Oncol Clin N Am 1999;8:307. [Crossref] [PubMed]
- Webb BD, Walsh GL, Roberts DB, et al. Primary tracheal malignant neoplasms: the University of Texas MD Anderson Cancer Center experience. J Am Coll Surg 2006;202:237-46. [Crossref] [PubMed]
- El Marjany M, Arsalane A, Sifat H, et al. Primary adenoid cystic carcinoma of the trachea: a report of two cases and literature review. Pan Afr Med J 2014;19:32. [Crossref] [PubMed]
- Stevic R, Milenkovic B. Tracheobronchial tumors. J Thorac Dis 2016;8:3401-13. [Crossref] [PubMed]
- Detterbeck FC. Management of carcinoid tumors. Ann Thorac Surg 2010;89:998-1005. [Crossref] [PubMed]
- Shepherd MP. Endobronchial metastatic disease. Thorax 1982;37:362-5. [Crossref] [PubMed]
- Kiryu T, Hoshi H, Matsui E. Endotracheal/endobronchial metastases: clinicopathologic study with special reference to developmental modes. Chest 2001;119:768-75. [Crossref] [PubMed]
- Blasco M, Quadrelli SA, Bosio M, et al. Synovial Sarcoma and Endobronchial Invasion. J Bronchology Interv Pulmonol 2008;15:167-9.
- Bilaçeroğlu S. Endobronchial Ablative Therapies. Clin Chest Med 2018;39:139-48. [Crossref] [PubMed]
- Israel RH, Poe RH. Hemoptysis. Clin Chest Med 1987;8:197-205. [PubMed]
- Jabbardarjani H, Herth F, Kiani A. Central Airway Obstruction Masquerading as Difficult-to-Treat Asthma: A Retrospective Study. J Bronchology Interv Pulmonol 2009;16:6-9. [Crossref] [PubMed]
- Hollingsworth HM. Wheezing and stridor. Clin Chest Med 1987;8:231-40. [PubMed]
- Mehta AC, Harris RJ, De Boer GE. Endoscopic management of benign airway stenosis. Clin Chest Med 1995;16:401-13. [PubMed]
- Brodsky JB. Bronchoscopic procedures for central airway obstruction. J Cardiothorac Vasc Anesth 2003;17:638-46. [Crossref] [PubMed]
- Bolliger CT, Sutedja TG, Strausz J. Therapeutic bronchoscopy with immediate effect: laser, electrocautery, argon plasma coagulation and stents. Eur Respir J 2006;27:1258-71. [Crossref] [PubMed]
- Collins J, Stern EJ. Chest Radiology: The Essentials. Wolters Kluwer Health: Philadelphia 2007.
- Santacruz JF, Mehta AC. Airway complications and management after lung transplantation: ischemia, dehiscence, and stenosis. Proc Am Thorac Soc 2009;6:79-93. [Crossref] [PubMed]
- Boiselle PM. Imaging of the large airways. Clin Chest Med 2008;29:181-93. [Crossref] [PubMed]
- Chung JH, Kanne JP. Multidetector-row Computed Tomography of Diffuse Tracheal Disease: Pictorial Review. J Bronchology Interv Pulmonol 2009;16:28-36. [Crossref] [PubMed]
- Whyte RI, Quint LE, Kazerooni EA, et al. Helical computed tomography for the evaluation of tracheal stenosis. Ann Thorac Surg 1995;60:27-30. [Crossref] [PubMed]
- Seijo LM, Sterman DH. Interventional pulmonology. N Engl J Med 2001;344:740-9. [Crossref] [PubMed]
- Jeon K, Kim H, Yu CM, et al. Rigid bronchoscopic intervention in patients with respiratory failure caused by malignant central airway obstruction. J Thorac Oncol 2006;1:319-23. [Crossref] [PubMed]
- Nihei K, Ishikura S, Kawashima M, et al. Short-course palliative radiotherapy for airway stenosis in non-small cell lung cancer. Int J Clin Oncol 2002;7:284-8. [PubMed]
- Guibert N, Mazieres J, Lepage B, et al. Prognostic factors associated with interventional bronchoscopy in lung cancer. Ann Thorac Surg 2014;97:253-9. [Crossref] [PubMed]
- Ernst A, Simoff M, Ost D, et al. Prospective risk-adjusted morbidity and mortality outcome analysis after therapeutic bronchoscopic procedures: results of a multi-institutional outcomes database. Chest 2008;134:514-9. [Crossref] [PubMed]
- Husain SA, Finch D, Ahmed M, et al. Long-term follow-up of ultraflex metallic stents in benign and malignant central airway obstruction. Ann Thorac Surg 2007;83:1251-6. [Crossref] [PubMed]
- Sergio C, Foccoli P, Toninelli C, et al. Nd:YAG Laser Therapy in Lung Cancer: An 11-Year Experience with 2,253 Applications in 1,585 Patients. J Bronchology Interv Pulmonol 1994;1:105-11.
- Ali MS, Sorathia L. Palliative Care and Interventional Pulmonology. Clin Chest Med. 2018;39:57-64. [Crossref] [PubMed]
- Simoff MJ, Lally B, Slade MG, et al. Symptom management in patients with lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:e455S-e497S.
- Lee P, Kupeli E, Mehta AC. Therapeutic bronchoscopy in lung cancer. Laser therapy, electrocautery, brachytherapy, stents, and photodynamic therapy. Clin Chest Med 2002;23:241-56. [Crossref] [PubMed]
- Folch E, Mehta AC. Airway interventions in the tracheobronchial tree. Semin Respir Crit Care Med 2008;29:441-52. [Crossref] [PubMed]
- Du Rand IA, Barber PV, Goldring J, et al. Summary of the British Thoracic Society guidelines for advanced diagnostic and therapeutic flexible bronchoscopy in adults. Thorax 2011;66:1014-5. [Crossref] [PubMed]
- Bolliger CT, Mathur PN, Beamis JF, et al. ERS/ATS statement on interventional pulmonology. European Respiratory Society/American Thoracic Society. Eur Respir J 2002;19:356-73. [PubMed]
- Ayers ML, Beamis JF Jr. Rigid bronchoscopy in the twenty-first century. Clin Chest Med 2001;22:355-64. [Crossref] [PubMed]
- Ernst A, Silvestri GA, Johnstone D, et al. Interventional pulmonary procedures: Guidelines from the American College of Chest Physicians. Chest 2003;123:1693-717. [Crossref] [PubMed]
- Alraiyes AH, Machuzak MS. Rigid bronchoscopy. Semin Respir Crit Care Med 2014;35:671-80. [Crossref] [PubMed]
- Yarmus L, Feller-Kopman D. Bronchoscopes of the twenty-first century. Clin Chest Med 2010;31:19-27. [Crossref] [PubMed]
- Dutau H, Vandemoortele T, Breen DP. Rigid bronchoscopy. Clin Chest Med 2013;34:427-35. [Crossref] [PubMed]
- Dumon JF, Reboud E, Garbe L, et al. Treatment of tracheobronchial lesions by laser photoresection. Chest 1982;81:278-84. [Crossref] [PubMed]
- Desai SJ, Mehta AC, VanderBrug Medendorp S, et al. Survival experience following Nd:YAG laser photoresection for primary bronchogenic carcinoma. Chest 1988;94:939-44. [Crossref] [PubMed]
- Cavaliere S, Venuta F, Foccoli P, et al. Endoscopic treatment of malignant airway obstructions in 2,008 patients. Chest 1996;110:1536-42. [Crossref] [PubMed]
- Wahidi MM, Herth FJ, Ernst A. State of the art: interventional pulmonology. Chest 2007;131:261-74. [Crossref] [PubMed]
- Reddy C, Majid A, Michaud G, et al. Gas embolism following bronchoscopic argon plasma coagulation: a case series. Chest 2008;134:1066-9. [Crossref] [PubMed]
- Seaman JC, Musani AI. Endobronchial ablative therapies. Clin Chest Med 2013;34:417-25. [Crossref] [PubMed]
- Coulter TD, Mehta AC. The heat is on: impact of endobronchial electrosurgery on the need for Nd-YAG laser photoresection. Chest 2000;118:516-21. [Crossref] [PubMed]
- Sheski FD, Mathur PN. Endobronchial electrosurgery: argon plasma coagulation and electrocautery. Semin Respir Crit Care Med 2004;25:367-74. [Crossref] [PubMed]
- Mahmood K, Wahidi MM. Ablative therapies for central airway obstruction. Semin Respir Crit Care Med 2014;35:681-92. [Crossref] [PubMed]
- Morice RC, Ece T, Ece F, et al. Endobronchial argon plasma coagulation for treatment of hemoptysis and neoplastic airway obstruction. Chest 2001;119:781-7. [Crossref] [PubMed]
- DiBardino DM, Lanfranco AR, Haas AR. Bronchoscopic Cryotherapy. Clinical Applications of the Cryoprobe, Cryospray, and Cryoadhesion. Ann Am Thorac Soc 2016;13:1405-15. [Crossref] [PubMed]
- Mathur PN, Wolf KM, Busk MF, et al. Fiberoptic bronchoscopic cryotherapy in the management of tracheobronchial obstruction. Chest 1996;110:718-23. [Crossref] [PubMed]
- Sheski FD, Mathur PN. Cryotherapy, electrocautery, and brachytherapy. Clin Chest Med 1999;20:123-38. [Crossref] [PubMed]
- Boujaoude Z, Young D, Lotano R, et al. Cryosurgery for the immediate treatment of acute central airway obstruction. J Bronchology Interv Pulmonol 2013;20:45-7. [Crossref] [PubMed]
- Schumann C, Hetzel M, Babiak AJ, et al. Endobronchial tumor debulking with a flexible cryoprobe for immediate treatment of malignant stenosis. J Thorac Cardiovasc Surg 2010;139:997-1000. [Crossref] [PubMed]
- Hetzel M, Hetzel J, Schumann C, et al. Cryorecanalization: a new approach for the immediate management of acute airway obstruction. J Thorac Cardiovasc Surg 2004;127:1427-31. [Crossref] [PubMed]
- Inaty H, Folch E, Berger R, et al. Unimodality and Multimodality Cryodebridement for Airway Obstruction. A Single-Center Experience with Safety and Efficacy. Ann Am Thorac Soc 2016;13:856-61. [Crossref] [PubMed]
- Vishwanath G, Madan K, Bal A, et al. Rigid bronchoscopy and mechanical debulking in the management of central airway tumors: an Indian experience. J Bronchology Interv Pulmonol 2013;20:127-33. [Crossref] [PubMed]
- Mathisen DJ, Grillo HC. Endoscopic relief of malignant airway obstruction. Ann Thorac Surg 1989;48:469-73. [Crossref] [PubMed]
- Hautmann H, Gamarra F, Pfeifer KJ, et al. Fiberoptic bronchoscopic balloon dilatation in malignant tracheobronchial disease: indications and results. Chest 2001;120:43-9. [Crossref] [PubMed]
- McArdle JR, Gildea T, Mehta AC. Balloon Bronchoplasty: Its Indications, Benefits, and Complications. J Bronchology Interv Pulmonol 2005;12:123-7.
- Kennedy MP, Morice RC, Jimenez CA, et al. Treatment of bronchial airway obstruction using a rotating tip microdebrider: a case report. J Cardiothorac Surg 2007;2:16. [Crossref] [PubMed]
- Lunn W, Bagherzadegan N, Munjampalli SK, et al. Initial Experience With a Rotating Airway Microdebrider. J Bronchology Interv Pulmonol 2008;15:91-4.
- Casal RF, Iribarren J, Eapen G, et al. Safety and effectiveness of microdebrider bronchoscopy for the management of central airway obstruction. Respirology 2013;18:1011-5. [Crossref] [PubMed]
- Folch E, Keyes C. Airway stents. Ann Cardiothorac Surg 2018;7:273-83. [Crossref] [PubMed]
- Wood DE, Liu YH, Vallières E, et al. Airway stenting for malignant and benign tracheobronchial stenosis. Ann Thorac Surg 2003;76:167-72. [Crossref] [PubMed]
- Lunn W, Feller-Kopman D, Wahidi M, et al. Endoscopic removal of metallic airway stents. Chest 2005;127:2106-12. [Crossref] [PubMed]
- Casal RF. Update in airway stents. Curr Opin Pulm Med 2010;16:321-8. [Crossref] [PubMed]
- Furukawa K, Ishida J, Yamaguchi G, et al. The role of airway stent placement in the management of tracheobronchial stenosis caused by inoperable advanced lung cancer. Surg Today 2010;40:315-20. [Crossref] [PubMed]
- Lee P, Kupeli E, Mehta AC. Airway Stents. Clin Chest Med 2010;31:141-50. [Crossref] [PubMed]
- Saji H, Furukawa K, Tsutsui H, et al. Outcomes of airway stenting for advanced lung cancer with central airway obstruction. Interact Cardiovasc Thorac Surg 2010;11:425-8. [Crossref] [PubMed]
- Razi SS, Lebovics RS, Schwartz G, et al. Timely airway stenting improves survival in patients with malignant central airway obstruction. Ann Thorac Surg 2010;90:1088-93. [Crossref] [PubMed]
- Mantovani G, Astara G, Manca G, et al. Endoscopic laser ablation as palliative treatment of endobronchial, nonresectable, or recurrent lung cancer: assessment of its impact on quality of life. Clin Lung Cancer 2000;1:277-85. [Crossref] [PubMed]
- Mahmood K, Wahidi MM, Thomas S, et al. Therapeutic bronchoscopy improves spirometry, quality of life, and survival in central airway obstruction. Respiration 2015;89:404-13. [Crossref] [PubMed]
- Ost DE, Ernst A, Grosu HB, et al. Therapeutic bronchoscopy for malignant central airway obstruction: success rates and impact on dyspnea and quality of life. Chest 2015;147:1282-98. [Crossref] [PubMed]
- Ost DE, Ernst A, Grosu HB, et al. Complications Following Therapeutic Bronchoscopy for Malignant Central Airway Obstruction: Results of the AQuIRE Registry. Chest 2015;148:450-71. [Crossref] [PubMed]
Cite this article as: Oberg C, Folch E, Santacruz JF. Management of malignant airway obstruction. AME Med J 2018;3:115.

