Etiopathogenesis of malignant pleural effusion
Introduction
Malignant pleural effusion (MPE) is featured by containing malignant cells (1,2). It is a frequent finding in patients with metastatic disease and it develops in 15% of patients with malignant disease (3-7), and its presence generally indicates a poor prognosis (8).
Two-thirds of all cases have a pleural effusion as one or sole initial manifestation of malignant disease. Primary tumors that most frequently develop MPE are lung, breast cancer, and lymphoma accounting for 75% of all cases. A MPE can develop in primary or metastatic malignancies of the pleura by spreading of malignant cells within the intrapleural cavity and into lymphatics causing their obstruction (1,2,5). The otherwise physiologic balance between the secretion of fluids into the pleural space and its reabsorption is largely disturbed by the occurrence of a MPE.
Advancements in molecular medicine enabled that the impact of tumor-host cell interactions has been recognized as an important mechanism in development of MPE.
Epidemiology of MPE
In adult population, 95% of MPEs develops from a metastatic site, and 75% of them originate from lung, breast cancer, and lymphoma (5), while the primary tumor remains unknown in approximately 5–6% of patients. During the course of malignant disease, nearly 50% of breast cancer patients have a MPE, approximately one-fourth of patients with lung cancer and one-third of patients with lymphoma, being most frequent malignancies followed by gynecological cancers and malignant mesothelioma (9,10) (Table 1).
Table 1
| Malignancy | Histologic subtype | Prevalence (%) |
|---|---|---|
| Lung cancer | Lung adenocarcinoma | 29–37 |
| Small cell carcinoma of the lung | 6–9 | |
| Breast cancer | Breast adenocarcinoma | 8–40 |
| Gynecological malignancy | Ovarian adenocarcinoma | 18–20 |
| Gastrointestinal cancer | Gastric adenocarcinoma | 2 |
| Colorectal | 1 | |
| Renal cell carcinoma | 1 | |
| Pancreatic adenocarcinoma | 3 | |
| Hematological malignancy | Lymphoma | 3–16 |
| Skin cancer | Melanoma | 5–6 |
| Mesothelioma | Malignant mesothelioma | 1–6 |
| Sarcoma | Sarcoma | 1–3 |
Summarized and modified after Clive et al. 2014 (10).
The MPE is the initial manifestation of a malignant disease in two-thirds of patients, with around 50% of them originating from lung cancer.
Although lung cancer subtypes have many similar histologic characteristics, there are many differences regarding their molecular features (11), squamous cell cancer being most frequent tumor developing a MPE in man, basically infiltrating directly the pleura. MPE is the initial sign of disease in 8–15% patients with lung cancer while in 40–50% it develops during lung cancer progression, and it is typically ipsilateral in 90% of those patients and bilateral or contralateral in 10% (12-14). The prevalence of MPE in SCLC is 10–38%, and unlike squamous cell lung cancer, it is the consequence of indirect infiltration of the lymphatics (15).
In breast cancer, the prevalence of MPE is 2–11%, most frequently one-sided, ipsilateral, most common in triple-negative breast cancer, and most often occurrence of the MPE is a bad prognostic factor. Interestingly, it can develop years after the diagnosis of breast cancer has been established. In breast cancer metastatic dissemination into the pleural space occurs via the lymphatic vessels (8,16,17). It has been noted that in the most invasive breast cancer subtype, triple-negative breast cancer, metastases develop most commonly between the second and third year after diagnosis been established. One of the characteristics of breast cancer metastases is that they often have subsequent mutations and molecular changes, so that is why Ki-67, a poor prognosis biomarker is determined in MPE, with increased values observed in 63% of MPE (16,17).
The most frequent (peritoneal) manifestation of epithelial ovarian cancer is MPE, recorded in 33–53% of cases, with ovarian cancer cells infiltrating directly the pleural structures directly via the diaphragm, pleuroperitoneal route or hematogenous dissemination (18). MPE occurs in 15% of newly diagnosed patients, as the initial clinical manifestation of ovarian cancer (18,19). Ipsilateral MPE is observed in 77%, while bilateral in 23%. Well recognized ovarian cancer biomarkers CA-125 and CA-15-3 are commonly found in increased levels in blood and pleural effusion as well (18).
When it comes to the MPE in Non-Hodgkin lymphoma, its prevalence is 16–20% of cases, that are more often left sided, most frequent in diffuse giant-cell B lymphoma (60%), and in follicular lymphoma (20%). The underlying pathophysiological mechanisms MPE develops via direct pleural infiltration with tumor-host cell interactions, lymph vessels obstruction with invasion of hilar and mediastinal lymph nodes, obstruction of the ductus thoracicus leading to chylothorax (20,21).
In Hodgkin lymphoma MPE develops as initial clinical manifestation of the disease in 10–30%, while in 60% of cases it occurs during further lymphoma progression. It should be noted that lymphomas are the most often cause of MPE occurrence in children and can also develop in patients with primary lymphoma of the pleura (20-22). Diagnosing MPE in lymphomas is rather difficult, a big challenge, mostly due to the paucity of cells in the fluid (21). MPE in lymphomas generally have poor prognosis as well, and moreover, around one third of lymphoma cases with MPE are chemotherapy-resistant (23,24).
Malignant pleural mesothelioma has prevalence of MPE of 54–90% of cases, commonly presenting at an early stage of disease (23,25).
It is important to underline that patients with a diagnosis of malignant disease can display a “paramalignant” pleural effusion because of local effects of their tumor. There are a variety of them such as atelectasis due to an intrabronchial obstruction, post-obstructive pneumonia with a parapneumonic effusion, some general tumor-related events like venous thromboembolism and hypoalbuminemia, and of course as adverse events of different treatment modalities such as radiotherapy and chemotherapy.
Pathophysiology and pathogenesis of MPE
An MPE can develop from primary or metastatic malignancies of the pleura by dissemination into pleural space and lymph vessels obstruction (1,2,5). The physiologic balance between influx of liquids into the pleural space and its reabsorption is largely disturbed producing a pleural effusion in a malignant disease, and both mechanisms, an increase in entry rate and a reduction in exit rate, contribute to development of MPE.
Malignant cells can invade the pleural space via the hematogenous, direct or lymphatic dissemination. Direct tumor involvement of pleura can lead to a pleural fluid accumulation by increasingly producing the liquid and thus influencing the normal parietal pleural lymphatic functioning. Tumor may extensively infiltrate pleural capillaries, leading to increased filtration, or may produce different cytokines that increase capillary permeability (26-28), while decreased plasma osmotic pressure or decreased pleural pressure can contribute to the enhanced entry of liquid as well. Elevations of hydrostatic pressure can thus also enlarge filtration from the pleural membrane microvessel.
On the other hand, tumor growth infiltrating the draining lymphatics or lymph nodes may block the lymphatic drainage thus decreasing the exit rate, the absorbtion rate of pleural fluid, with the subsequent accumulation of fluid in the pleural space (1), while different extrinsic factors including limited respiratory mobility, mechanical compression of lymph vessels with blockage of their stomata, may be responsible in the cases when lymphatics activity is significantly damaged, but not due to direct damage of the vessels (29). In some cases of lymphatic infiltration, the decrease in the exit rate may represent a key mechanism of MPE development, when the effusions can resolve after mediastinal irradiation of involved lymph nodes. In certain MPEs, extrapleural involvement of draining lymphatics may be the sole mechanism of effusion formation, that may explain the transudative type of malignant effusions, which is noted in about 10 percent of patients with MPE (30). Extensive tumor cells infiltration of pleural capillaries most probably may explain cases with rapid entry rates, which can be recognized clinically because of the rapid effusion reaccumulation after drainage or having high chest tube drainage rate.
An interesting observation is that only 55–60% of patients with metastases into pleura or lymph vessels and/or nodes have MPE (31), the reason is not quite clear, but there is evidence of more often poor outcome in those with “wet” pleural carcinosis in comparison to “dry” pleural carcinosis (31).
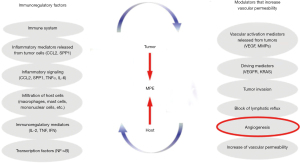
With the advancements in molecular medicine, the impact of tumor-host cell interactions has been recognized as an important pathogenetic mechanism in development of MPE, with the hyperproduction of pleural fluid from hyperpermeable vessels, the process recognized as a very important but complex mechanism in development of MPE. Variety of cells and molecules are part of this complex process, producing diversity of effects regarding pleural inflammation, tumor angiogenesis and vascular hyperpermeability. Tumor- and host-derived factors involved in MPE development include numerous secreted mediators: Osteopontin (OPN; secreted phosphoprotein 1), C-C motif chemokine ligand 2 (CCL2; monocyte chemoattractant protein-1), vascular endothelial growth factor (VEGF), tumor necrosis factor (TNF), Angiopoietins 1 and 2, Interleukin-5, Interleukin-6 etc. Some of them stimulate the pleural inflammation and include interleukin 2 (IL2), tumor necrosis factor (TNF) and interferon (INF); molecules that stimulate tumor angiogenesis appear to be angiopoietin 1 (ANG-1), angiopoietin 2 (AGN-2), while the molecules affecting vascular hyperpermeability include vascular endothelial growth factor (VEGF) that increase capillary permeability, matrix metalloproteinases (MMP), chemokine (c-c motif) ligand 2 (CCL), osteopontin (OPN), etc. (32-42). It has been demonstrated that mastocytes have an important contribution on inducing MPE as the key cells producing cytokines, such as tryptase alpha/beta 1 (AB1) and interleukin-1β (IL1 beta), leading to increased permeability (42). The secretion of tryptase alpha/beta 1 and interleukin-1β enhances the permeability of the pulmonary vessels and have the profound effect on activating the NF-ffB transcription factor, which fosters the accumulation of effusion and tumor progression (1,42) (Figure 1).
To summarize, in primary or metastatic pleural tumors, the balance between vasoactive mediators (e.g., VEGF, TNF, CCL2, OPN, etc.) and possibly protection molecules (e.g., endostatin) within the pleural cavity determines the process of vasoactive signaling with consequent development of pleural effusion. This very combination of signals is a key process dictating further host cell activation and recruitment. On the other hand, resident and incoming host cells exhibit multiple active roles, such as directly affecting malignant cells (transcription factor stimulation; rejection, tumor promotion, immunoediting and/or tumor escape), as well as producing some indirect effects on the pleural vessels, immune cell populations, and mesothelium, thus impacting inflammation, angiogenesis, vascular leakage, and/or intrapleural metastasis with development of new malignant foci within pleura (31) (Figure 2).
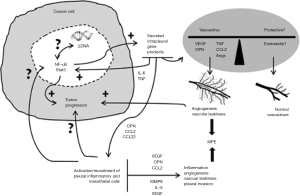
The genomic analysis of malignant cells detected that cancers with activating mutations EGFR, KRAS, PIK3CA, BRAF, MET, EML4/ALK and RET demonstrated significantly more frequently development of MPE (43-45), with evidence of different mutations in the primary tumor vs. pleural metastases (46-48).
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Dragan Subotic) for the series “Malignant Pleural Effusion” published in AME Medical Journal. The article has undergone external peer review.
Conflicts of Interest: The author has completed the ICMJE uniform disclosure form (available at https://amj.amegroups.com/article/view/10.21037/amj-2019-mpe-05/coif). The series “Malignant Pleural Effusion” was commissioned by the editorial office without any funding or sponsorship. The author has no other conflicts of interest to declare.
Ethical Statement:
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Psallidas I, Kalomenidis I, Porcel JM, et al. Malignant pleural effusion: From bench to bedside. Eur Respir Rev 2016;25:189-98. [Crossref] [PubMed]
- American Thoracic Society. Management of malignant pleural effusions. Am J Resp Crit Care Med 2000;162:1987-2001. [Crossref] [PubMed]
- Heffner JE, Klein JS. Recent advances in the diagnosis and management of malignant pleural effusions. Mayo Clin Proc 2008;83:235. [Crossref] [PubMed]
- Ferreiro L, Suárez-Antelo J, Valdés L. Pleural procedures in the management of malignant effusions. Ann Thorac Med 2017;12:3. [Crossref] [PubMed]
- American Society of Clinical Oncology. Malignant pleural effusion. Available online: http://www.cancer.net/navigating-cancer-care/side-effects/fluid-around-lungs-or-malignant-pleural-effusion
- Bibby AC, Dorn P, Psallidas I, et al. ERS/EACTS statement on the management of malignant pleural effusions. Eur J Cardiothorac Surg 2019;55:116-32. [Crossref] [PubMed]
- Feller-Kopman DJ, Reddy CB, DeCamp MM, et al. Management of Malignant Pleural Effusions. An Offcial ATS/STS/STR Clinical Practice Guideline. Am J Respir Crit Care Med 2018;198:839-49. [Crossref] [PubMed]
- Porcel JM, Solé C, Salud A, Bielsa S. Prognosis of Cancer with Synchronous or Metachronous Malignant Pleural Effusion. Lung 2017;195:775-9. [Crossref] [PubMed]
- Penz E, Watt KN, Hergott CA, et al. Management of malignant pleural effusion: Challenges and solutions. Cancer Manag Res 2017;9:229-41. [Crossref] [PubMed]
- Clive AO, Kahan BC, Hooper CE, et al. Predicting survival in malignant pleural effusion: Development and validation of the LENT prognostic score. Thorax 2014;69:1098-104. [Crossref] [PubMed]
- Inamura K. Lung Cancer: Understanding Its Molecular Pathology and the 2015 WHO Classification. Front Oncol 2017;7:193. [Crossref] [PubMed]
- Porcel JM, Bielsa S, Civit C, et al. Clinical features and survival of lung cancer patients with pleural effusions. Respirology 2015;20:654-9. [Crossref] [PubMed]
- Porcel JM. Malignant pleural effusions because of lung cancer. Curr Opin Pulm Med 2016;22:356-61. [Crossref] [PubMed]
- Cattapan K, Tanomkiat W, Geater SL, et al. Procedure-related tumour seeding in lung cancer with malignant pleural effusion: Radiological features and outcomes. J. Med. Imaging Radiat Oncol 2018;62:619-24. [Crossref] [PubMed]
- Ryu JS, Lim JH, Lee JM, et al. Minimal Pleural Effusion in Small Cell Lung Cancer: Proportion, Mechanisms, and Prognostic Effect. Radiology 2016;278:593-600. [Crossref] [PubMed]
- Shinohara T, Yamada H, Fujimori Y, et al. Malignant pleural effusion in breast cancer 12 years after mastectomy that was successfully treated with endocrine therapy. Am J Case Rep 2013;14:184-7. [Crossref] [PubMed]
- Soni A, Ren Z, Hameed O, et al. From Breast Cancer Subtypes Predispose the Site of Distant Metastases. Am J Clin Pathol 2015;143:471-8. [Crossref] [PubMed]
- Porcel JM, Diaz JP, Chi DS. Clinical implications of pleural effusions in ovarian cancer. Respirology 2012;17:1060-7. [Crossref] [PubMed]
- OʼLeary BD, Treacy T, Geoghegan T, et al. Incidental Thoracic Findings on Routine Computed Tomography in Epithelial Ovarian Cancer. Int J Gynecol Cancer 2018;28:1073-6. [Crossref] [PubMed]
- Sun ML, Shang B, Gao JH, et al. Rare case of primary pleural lymphoma presenting with pleural effusion. Thorac Cancer 2016;7:145-50. [Crossref] [PubMed]
- Choy CF, Lee S. Pleural lymphoma. J Bronchol Interv Pulmonol 2016;23:146-8. [Crossref] [PubMed]
- Alexandrakis MG, Passam FH, Kyriakou DS, et al. Pleural effusions in hematologic malignancies. Chest 2004;125:1546-55. [Crossref] [PubMed]
- Lepus CM, Vivero M. Updates in Effusion Cytology. Surg Pathol Clin 2018;11:523-44. [Crossref] [PubMed]
- Chen YP, Huang HY, Lin KP, et al. Malignant effusions correlate with poorer prognosis in patients with diffuse large B-cell Lymphoma. Am J Clin Pathol 2015;143:707-15. [Crossref] [PubMed]
- Cheah HM, Lansley SM, Varano della Vergiliana JF, et al. Malignant pleural fluid from mesothelioma has potent biological activities. Respirology 2017;22:192-9. [Crossref] [PubMed]
- Saffran L, Ost DE, Fein AM, et al. Outpatient pleurodesis of malignant pleural effusions using a small-bore pigtail catheter. Chest 2000;118:417. [Crossref] [PubMed]
- Bazerbashi S, Villaquiran J, Awan MY, et al. Ambulatory intercostal drainage for the management of malignant pleural effusion: a single center experience. Ann Surg Oncol 2009;16:3482. [Crossref] [PubMed]
- Davies HE, Mishra EK, Kahan BC, et al. Effect of an indwelling pleural catheter vs chest tube and talc pleurodesis for relieving dyspnea in patients with malignant pleural effusion: the TIME2 randomized controlled trial. JAMA 2012;307:2383. [Crossref] [PubMed]
- Yalcin NG, Choong CKC, Eizenberg N. Anatomy and Pathophysiology of the Pleura and Pleural Space. Thorac Surg Clin 2013;23:1-10. [Crossref] [PubMed]
- Demmy TL, Gu L, Burkhalter JE, et al. Optimal management of malignant pleural effusions (results of CALGB 30102). J Natl Compr Canc Netw 2012;10:975. [Crossref] [PubMed]
- Stathopoulos GT, Kalomenidis I. Malignant pleural effusion: Tumor-host interactions unleashed. Am J Respir Crit Care Med 2012;186:487-92. [Crossref] [PubMed]
- Cui R, Takahashi F, Ohashi R, et al. Osteopontin is involved in the formation of malignant pleural effusion in lung cancer. Lung Cancer 2009;63:368-74. [Crossref] [PubMed]
- Psallidas I, Stathopoulos GT, Maniatis NA, et al. Secreted phosphoprotein-1 directly provokes vascular leakage to foster malignant pleural effusion. Oncogene 2013;32:528-35. [Crossref] [PubMed]
- Stathopoulos GT, Psallidas I, Moschos C, et al. A central role for tumor-derived monocyte chemoattractant protein-1 in malignant pleural effusion. J Natl Cancer Inst 2008;100:1464-76. [Crossref] [PubMed]
- Yano S, Shinohara H, Herbst RS, et al. Production of experimental malignant pleural effusions is dependent on invasion of the pleura and expression of vascular endothelial growth factor/vascular permeability factor by human lung cancer cells. Am J Pathol 2000;157:1893-903. [Crossref] [PubMed]
- Ishii H, Yazawa T, Sato H, et al. Enhancement of pleural dissemination and lymph node metastasis of intrathoracic lung cancer cells by vascular endothelial growth factors (VEGFs) Lung Cancer 2004;45:325-37. [Crossref] [PubMed]
- Stathopoulos GT, Kollintza A, Moschos C, et al. Tumor necrosis factor-α promotes malignant pleural effusion. Cancer Res 2007;67:9825-34. [Crossref] [PubMed]
- Moschos C, Psallidas I, Kollintza A, et al. The angiopoietin/Tie2 axis mediates malignant pleural effusion formation. Neoplasia 2009;11:298-304. [Crossref] [PubMed]
- Stathopoulos GT, Sherrill TP, Karabela SP, et al. Host-derived interleukin-5 promotes adenocarcinoma-induced malignant pleural effusion. Am J Respir Crit Care Med 2010;182:1273-81. [Crossref] [PubMed]
- Yeh HH, Lai WW, Chen HHW, et al. Autocrine IL-6-induced Stat3 activation contributes to the pathogenesis of lung adenocarcinoma and malignant pleural effusion. Oncogene 2006;25:4300-9. [Crossref] [PubMed]
- Chen Y, Mathy NW, Lu H. The role of VEGF in the diagnosis and treatment of Malignant pleural effusion in patients with non-small cell lung cancer Mol Med Rep 2018;17:8019-30. (review). [Crossref] [PubMed]
- Giannou AD, Marazioti A, Spella M, et al. Mast cells mediate malignant pleural effusion formation. J. Clin. Investig 2015;125:2317-34. [Crossref] [PubMed]
- Network CGAR. Comprehensive molecular profiling of lung adenocarcinoma. Nature 2014;511:543-50. [Crossref] [PubMed]
- Agalioti T, Giannou AD, Krontira AC, et al. Mutant KRAS promotes malignant pleural effusion formation. Nat Commun 2017;8:15205. [Crossref] [PubMed]
- Wu SG, Gow CH, Yu CJ, et al. Frequent epidermal growth factor receptor gene mutations in malignant pleural effusion of lung adenocarcinoma. Eur Respir J 2008;32:924-30. [Crossref] [PubMed]
- Spella M, Giannou AD, Stathopoulos GT. Switching off malignant pleural effusion formation-fantasy or future? J Thorac Dis 2015;7:1009-20. [PubMed]
- Carter J, Miller JA, Feller-Kopman D, et al. Molecular profiling of malignant pleural effusion in metastatic non-small-cell lung carcinoma the effect of preanalytical factors. Ann Am Thorac Soc 2017;14:1169-76. [PubMed]
- Han HS, Eom DW, Kim JH, et al. EGFR mutation status in primary lung adenocarcinomas and corresponding metastatic lesions: discordance in pleural metastases. Clin Lung Cancer 2011;12:380-6. [Crossref] [PubMed]
Cite this article as: Jovanovic D. Etiopathogenesis of malignant pleural effusion. AME Med J 2021;6:28.