Robotic-assisted thoracoscopic surgery: left upper lobectomy
Clinical data
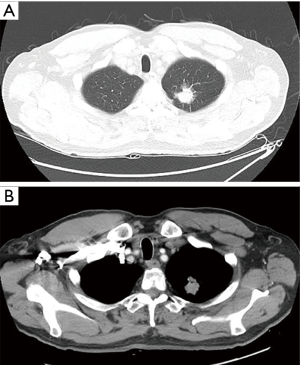
The patient was a 63-year-old man admitted because of cough and pulmonary nodule detected by computed tomography (CT). Chest CT (Figure 1) showed a mass located in the posterior segment of the left upper lobe (2.5 cm × 2 cm) that was lobulated with burr-like edges. The mass did not change after anti-inflammation therapy. The positron emission tomography-CT SUVmax was 6.5 and malignancy was considered. The mediastinal lymph node was negative and there was no distant metastasis. The patient’s complaints did not include chills, low fever, night sweats, hoarseness, or fatigue of upper limbs. The cardiopulmonary function, blood gas analysis, and laboratory tests were normal. There was no positive sign or supraclavicular lymph node enlargement on physical examination. He had no past medical history. Preoperative stage was cT1N0M0 (IA). Informed consent for robotic-assisted thoracic lobectomy was obtained from patient before operation.
Operation steps
Anesthesia and body position
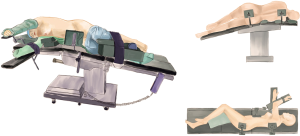
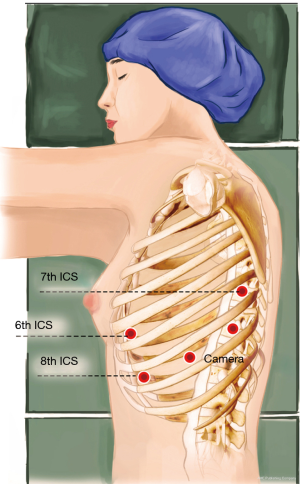
After the induction of general anesthesia, the patient was placed in a right lateral decubitus position under double-lumen endotracheal intubation. With his hands placed in front of his head, the patient was fixed in a jackknife position with single-lung (right) ventilation (Figure 2).
Ports
A 1.5-cm camera port (for a 12-mm trocar) was created in the 8th intercostal space (ICS) at the left mid axillary line, and three separate 1.0-cm working ports (for 8-mm trocars) were made in the 6th ICS (#1 arm) at the left anterior axillary line, the 8th ICS (#2 arm) at the left posterior axillary line, and the left 7th ICS (#3 arm), 2 cm from the spine. An auxiliary port (for a 12-mm trocar) was made in the 8th ICS near the costal arch (Figure 3).
Installation of the surgical arms
The robot patient cart was positioned directly above the operating table and then connected. The #2 arm was connected to a bipolar cautery forces, and the #1 arm was connected to a unipolar cautery hook. An incision protector was used in the auxiliary port.
Surgical procedure
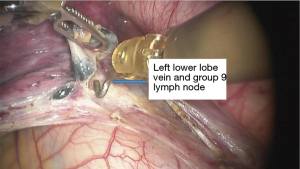
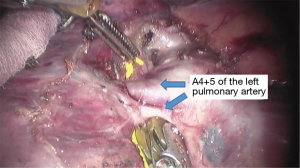
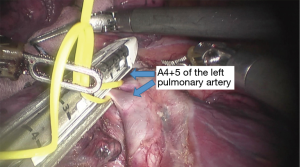
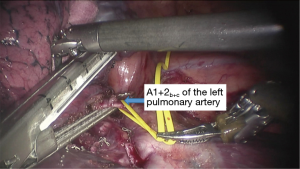
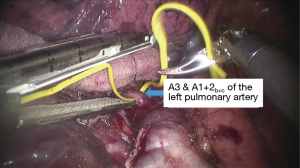
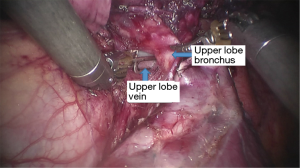
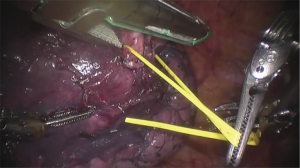
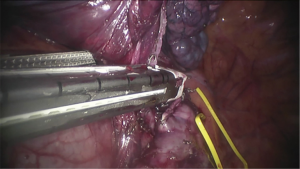
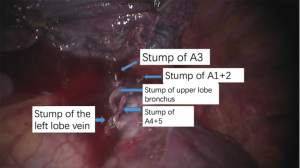
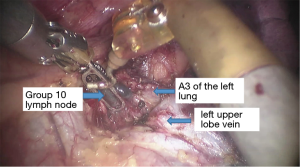
See Figures 4,5,6,7,8,9,10,11,12,13,14,15,16,17,18.
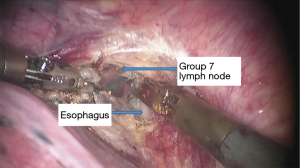
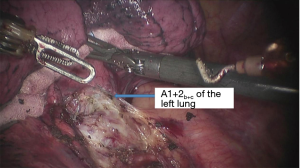
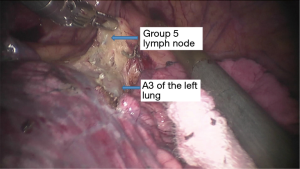
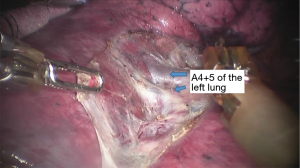
Postoperative condition
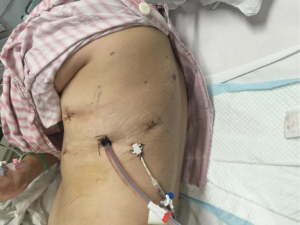
Postoperative treatments included anti-inflammatory and phlegm-resolving treatments. The thoracic drainage tube was withdrawn 1 day after surgery, and the patient was discharged 3 days after surgery. No complications were observed during hospitalization. Pathological diagnosis was left upper lobe invasive adenocarcinoma. All lymph nodes were negative. Postoperative pathological stage was pT1aN0M0 (IA adenocarcinoma).
Discussion
Originally, the robotic system was developed for cardiac surgery. The first internal mammary artery graft were performed in 1999 and 2000 (1,2). After these experiences, robotic systems were used in other fields, such as thoracic surgery for a wide range of procedures, starting with simple ones such the resection of anterior or posterior mediastinal masses (3-5). Robotic technology has certain advantages, especially in minimally invasive anatomic lung resection. The advantages of the robot compared with video-assisted thoracoscopic surgery include improved visualization, improved instrumentation that provides the surgeon with more degrees of movement, better lymph node visualization and dissection, high magnification, the ability to teach using a dual console, and a simulator (6). The left upper lobectomy is the most challenging of all lobectomies because of the complex arterial branches in the left upper lobe. In particular, the short branch A3 hemorrhages easily when dissociating and pulling. The da Vinci Surgical System has four mechanical arms. The #3 arm can help the surgeon find the pulling location, which reduces the assistant’s work. Hole of the #3 arm is relatively easy bleeding holes, beginners need to look at the punch. In all surgeries, we dissect the hilum using the posterior approach to expose the pulmonary artery which improves the safety. We inject CO2 before withdrawing the specimen of lung lobe to form a closed space, which can keep the operation field clear. The assistant uses a forceps tong with a gauze roll to pull the lobe to provide room for the surgeon. Straight nails are used to cut the vessels to lower the cost for the patients.
Acknowledgements
Funding: None.
Footnote
Conflicts of Interest: The authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/amj.2017.01.10). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Carpentier A, Loulmet D, Aupecle B, et al. Computer-assisted cardiac surgery. Lancet 1999;353:379-80. [Crossref] [PubMed]
- Kappert U, Cichon R, Gulielmos V, et al. Robotic-enhanced Dresden technique for minimally invasive bilateral internal mammary artery grafting. Heart Surg Forum 2000;3:319-21. [PubMed]
- Yoshino I, Hashizume M, Shimada M, et al. Thoracoscopic thymomectomy with the da Vinci computer-enhanced surgical system. J Thorac Cardiovasc Surg 2001;122:783-5. [Crossref] [PubMed]
- Bodner J, Wykypiel H, Greiner A, et al. Early experience with robot-assisted surgery for mediastinal masses. Ann Thorac Surg 2004;78:259-65; discussion 265-6. [Crossref] [PubMed]
- Rea F, Marulli G, Bortolotti L, et al. Experience with the "da Vinci" robotic system for thymectomy in patients with myasthenia gravis: report of 33 cases. Ann Thorac Surg 2006;81:455-9. [Crossref] [PubMed]
- Cerfolio RJ, Bryant AS. Perspectives on robotic pulmonary resection: It's current and future status. Ann Cardiothorac Surg 2012;1:59-60. [PubMed]
Cite this article as: Xiang J, Yang S, Guo W, Jin R, Zhang Y, Chen X, Wu H, Du H, Han D, Chen K, Li H. Robotic-assisted thoracoscopic surgery: left upper lobectomy. AME Med J 2017;2:22.