
Biopsy: CT-guided fine needle aspiration is best? radiologist perspective
Introduction
The recent and increasing availability of computed tomography (CT) and the advances in lung screening techniques, with high resolution images and multidetector devices, with faster exam acquisition time and more sensitive images, allowed the identification of a vertiginously growing number of pulmonary nodules, and ever smaller lesions (1).
Given the increasing array of identified nodules, the major challenge for radiologists, pulmonologists, and thoracic surgeons is the ability to differentiate between benign and malignant lesions, and then listing the patients that can be followed and the others that need to be treated (2,3). Early treatment is always the focus, and the characterization of a malignant nodule, when still small, greatly increases the chances of effective and curative treatment (2,3).
Some lung lesions may be considered benign when a previous imaging examination suggests stability. However, lesions with specific tomographic characteristics that suggest malignancy require further investigation. Follow-up is a possibility in many cases, especially of patients with small nodules, however, this approach can often generate anxiety in the patient and can also delay early-stage treatment of the disease. In addition, it is important to consider that the follow-up CT uses ionizing radiation and requires a longer period of time for a diagnostic conclusion, considering the necessary time interval (4-6).
In the above-mentioned context, cytological or tissue sampling of suspected nodules by percutaneous guided imaging, more frequently CT, has increasingly been placed as an effective and safe alternative in the solution of the doubt of the nodule composition.
Discussion
The two most frequently used options in the percutaneous diagnosis of suspicious pulmonary nodules are fine needle aspiration biopsy (FNAB) and core (thick needle) biopsies. Both mentioned techniques present indications, limitations and characteristic risks, and both the applicant and the interventional radiologist must know these factors when indicating or performing one or the other procedure.
With respect to FNAB, this technique is often used in a number of situations in which malignancy is suspected. Thyroid nodules are a classic and successful example of performing percutaneous aspiration biopsies. Surely, thyroid nodules are the ones in which are most often performed fine needle punctures. In the last years, a significant improvement in the parameters of classification of thyroid nodules, such as the TIRADS classification, captained with small methodological differences among themselves, but with the similar proposal, by some groups in the world literature, provided a precise indication of puncture for those nodules with a higher risk of malignancy. The recent development of TIRADS classification by American College of Radiologists (ACR) confirmed this increasing need for adequate classification of thyroid lesions for precise indication to FNAB.
Solid, cystic or mixed nodular mammary lesions are also frequent targets of FNAB, in this case following the older and consecrated BIRADS classification, which elutes the nodules to be biopsied, separating them from those that can simply be followed with clinical and imaging follow-up, most often by ultrasound (US).
Increased lymph nodes with suspicious characteristics are also frequent targets of FNAB, with cervical, inguinal and axillary lymph nodes being the most frequently punctured.
As far as pulmonary nodules are concerned, aspiration puncture also plays a role, although today the indication of this technique is increasingly restricted. About 10 years ago, fine needle biopsies were much more frequent, while the percutaneous access approach gained its place within the arsenal of diagnostic possibilities. At this time, when surgical biopsies were still dominant, the possibility of fine-needle CT-guided diagnosis, without the risks of a surgical intervention, began to gain form, and became well accepted (7-9).
Percutaneous CT-guided FNAB is widely accepted as an accurate and safe procedure for the characterization of pulmonary nodules. Diagnostic accuracy rates and sensitivity reaches approximately 95%, but several studies reported a decrease in diagnostic accuracy for smaller lesions, ranging from 52% to 88%, with great variation between the studies (10) (Figure 1).
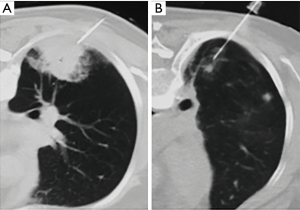
One inherent feature of the technique, which has limited it, is the small sampling of material that fine needle aspiration achieves. By its nature, samples obtained by FNAB are naturally cytological and, therefore, show high false negative indices. The need for more accurate sampling, with histological evaluation, was necessary (10).
Core biopsy has progressively gained space, as it presents higher accuracy, with low false negative indices, high positivity and similar complication índices, such as pneumothorax. The rates of parenchymal bleeding, in spite of larger needle calibers, did not represent a significant increase in the morbimortality of the procedure (11-18) (Figure 2).
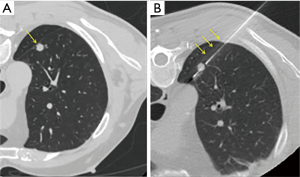
Pneumothorax is the most frequently complication in core biopsies of pulmonary nodules, with an incidence between 21% and 43% in the literature (19,20). Higher rates of pneumothorax, up to 65% in the literature, are observed for lesions of 10 mm or less. The percentage, however, of pneumothorax requiring percutaneous drainage varies from 4% to 35%. Fatal complications, like systemic arterial embolism and pulmonary hemorrhage, have been reported but are extremely rare (14,15,18,21,22).
Given the two percutaneous diagnostic possibilities listed, an issue that applies both to those who indicate the procedure—clinicians and surgeons, and who performs it—especially interventionists, is when to indicate one or another technique.
Main advantages of FNAB are the lowest cost of the material and complications indices. In contrast, the accuracy of the method is quite variable, attending frequently with high false negative indices or insufficient samplings.
Core biopsy, on the other hand, shows greater material sampling, with higher accuracy and less false negatives. In contrast, complication rates are higher, in despite of not leading to a significant increase in morbidity and mortality of the procedure. The cost is slightly higher if compared to FNAB, because of the material and the possible need of sedation or even general anesthesia (21,23,24).
It is a consensus between the majority of interventional radiologists who actually perform percutaneous biopsies, that the procedure should be made using thick needles, given the safety and accuracy of the core biopsy, with similar complications indices in relation to FNAB. Nowadays, this method is already well established, accepted as effective and safe by most services, with good acceptance even by more enlightened patients. The aspiration puncture may still be used, especially in larger nodules, with a higher risk of false negative samples, and in a group of patients at high risk of bleeding.
Conclusions
Most important, however, at the end of the above-mentioned considerations, is the consolidation currently tried by the interventionist practice as the first option in the diagnostic approach of suspected pulmonary nodules. Given the differences in techniques and outcomes between FNAB and core biopsy, both strategies have a minimal morbi-mortality rate if compared to conventional surgical biopsy, allowing rapid recovery after the procedure, with the majority of patients discharged asymptomatic at the same day, and also with lower overall costs. In addition, knowledge of the histological type of the malignant nodule before surgery allows a safer planning of the best surgical strategy, better access and the need of surgery extension.
In the understanding of the interventionist radiologists, the multidisciplinary practice, the exchange of information among the clinical teams that conduct the patient evaluation, brings unequivocal benefits, adding efforts for the earlier and more accurate diagnosis, and for an increasingly less invasive and resolutive treatment.
Acknowledgements
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Ricardo Sales dos Santos, Myrna Godoy, Juliana Franceschini and Hiran C. Fernando) for the series “Update on Lung Cancer Screening and the Management of CT Screening Detected Pulmonary Nodules” published in AME Medical Journal. The article has undergone external peer review.
Conflicts of Interest: The authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/amj.2017.11.06). The series “Update on Lung Cancer Screening and the Management of CT Screening Detected Pulmonary Nodules” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Henschke CI, McCauley DI, Yankelevitz DF, et al. Early lung cancer action project: overall design and findings from baseline screening. Lancet 1999;354:99-105. [Crossref] [PubMed]
- Diederich S, Wormanns D, Semik M, et al. Screening for early lung cancer with low-dose spiral CT: prevalence in 817 asymptomatic smokers. Radiology 2002;222:773-81. [Crossref] [PubMed]
- Henschke CI, Naidich DP, Yankelevitz DF, et al. Early lung cancer action project: initial finding on repeat screenings. Cancer 2001;92:153-9. [Crossref] [PubMed]
- Libby DM, Smith JP, Attorki NK, et al. Managing the small pulmonary nodule discovered by CT. Chest 2004;125:1522-9. [Crossref] [PubMed]
- MacMahon H, Austin JH, Gamsu G, et al. Guidelines for management of small pulmonary nodules detected on CT scans: a statement from the Fleischner society. Radiology 2005;237:395-400. [Crossref] [PubMed]
- Naidich DP, Bankier AA, MacMahon H, et al. Recommendations for the management of subsolid pulmonary nodules detected at CT: a statement from the Fleischner society. Radiology 2013;266:304-17. [Crossref] [PubMed]
- Gupta S, Wallace MJ, Cardella JF, et al. Quality improvement guidelines for percutaneous needle biopsy. J Vasc Interv Radiol 2010;21:969-75. [Crossref] [PubMed]
- Hiraki T, Miruma H, Gobara H, et al. CT fluoroscopy-guided biopsy of 1,000 pulmonary lesions performed with 20-gauge coaxial cutting needles: diagnostic yield and risck factors for diagnostic failure. Chest 2009;136:1612-7. [Crossref] [PubMed]
- Tsukada H, Satou T, Iwashima A, et al. Diagnostic accuracy of CT-guided automated needle biopsy of lung nodules. AJR Am J Roentgenol 2000;175:239-43. [Crossref] [PubMed]
- Takeshita J, Masago K, Kato R, et al. CT-guided fine-needle aspiration and core needle biopsies of pulmonary lesions: a single-center experience with 750 biopsies in Japan. AJR Am J Roentgenol 2015;204:29-34. [Crossref] [PubMed]
- Laurent F, Latrabe V, Vergier B, et al. CT-guided transthoracic needle biopsy of pulmonary nodules smaller than 20 mm: results with an automated 20-gauge coaxial cutting needle. Clin Radiol 2000;55:281-7. [Crossref] [PubMed]
- Choi SH, Chae EJ, Kim JE, et al. Percutaneous CT-guided aspiration and core biopsy of pulmonary nodules smaller than 1 cm: analysis of outcomes of 305 procedures from a tertiary referral center. AJR Am J Roentgenol 2013;201:964-70. [Crossref] [PubMed]
- Yamauchi Y, Izumi Y, Nakatsuka S, et al. Diagnostic performance of percutaneous core-needle lung biopsy under CT scan fluoroscopic guidance for pulmonary lesions measuring ≤10 mm. Chest 2011;140:1669-70. [Crossref] [PubMed]
- Jae LI, June IH, Miyeon Y, et al. Percutaneous core needle biopsy for small (≤10 mm) lung nodules: accurate diagnosis and complication rates. Diagn Interv Radiol 2012;18:527-30. [PubMed]
- Li Y, Du Y, Yang HF, et al. CT-guided percutaneous core needle biopsy for small (≤20 mm) pulmonary lesions. Clin Radiol 2013;68:e43-8. [Crossref] [PubMed]
- Yoshimura N, Takeda K, Tada H, et al. The factors determining diagnostic accuracy in CT-guided percutaneous needle biopsy of small pulmonary nodules. Nihon Kokyuki Gakkai Zasshi 2002;40:101-5. [PubMed]
- Hayashi N, Sakai T, Ktagawa M, et al. CT-guided biopsy of pulmonary nodules less than 3 cm: usefulness of the spring-operate core biopsy needle and frozen-section pathologic diagnosis. AJR Am J Roentgenol 1998;170:329-31. [Crossref] [PubMed]
- Choo JY, Park CM, Lee NK, et al. Percutaneous transthoracic needle biopsy of small (≤1 cm) lung nodules under C-arm cone-beam CT virtual navigation guidance. Eur Radiol 2013;23:712-9. [Crossref] [PubMed]
- Yeow KM, Tsay PK, Cheung YC, et al. Factors affecting diagnostic accuracy of CT-guided coaxial cutting needle lung biopsy: retrospective analysis of 631 procedures. J Vasc Interv Radiol 2003;14:581-8. [Crossref] [PubMed]
- Lee SM, Park CM, Lee KH, et al. C-arm cone-beatm CT-guided percutaneous transthoric needle biopsy of lung nodules: clinical experience in 1108 patients. Radiology 2014;271:291-300. [Crossref] [PubMed]
- Laurent F, Latrabe V, Vergier B, et al. Percutaneous CT-guided biopsy of the lung: comparison between aspiration and automated cutting needles using a coaxial technique. Cardiovasc Intervent Radiol 2000;23:266-72. [Crossref] [PubMed]
- Rocha RD, Azevedo AA, Falsarella PM, et al. Cerebral air embolism during CT-guided lung biopsy. Thorax 2015;70:1099-100. [Crossref] [PubMed]
- Kothary N, Lock L, Sze DY, et al. Computed tomography-guided percutaneous needle biopsy of pulmonary nodules: impact of nodule size on diagnostic accuracy. Clin Lung Cancer 2009;10:360-3. [Crossref] [PubMed]
- Hwang HS, Chung MJ, Lee JW, et al. C-arm cone-beam CT-guided percutaneous transthoracic lung biopsy: usefulness in evaluation of small pulmonary nodules. AJR Am J Roentgenol 2010;195:W400-7. [Crossref] [PubMed]
Cite this article as: Rahal Junior A, Falsarella PM, Santos RS, Mendes GF, Martins DLN, Garcia RG. Biopsy: CT-guided fine needle aspiration is best? radiologist perspective. AME Med J 2017;2:177.

